Deep-learning-enabled online mass spectrometry of the reaction product of a single catalyst nanoparticle
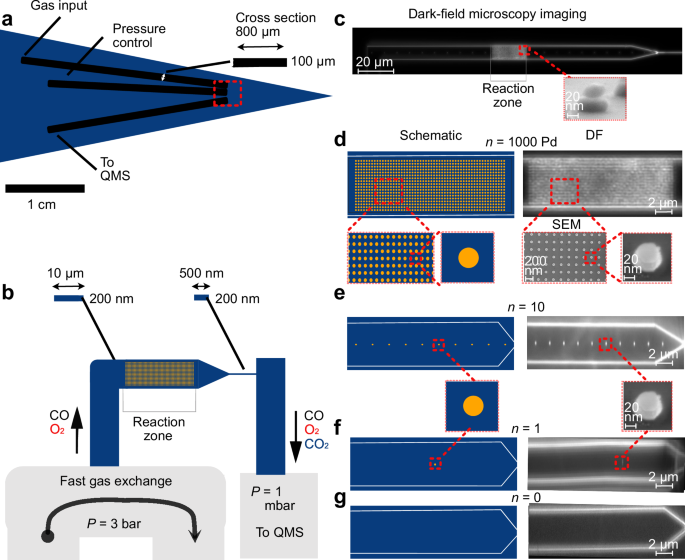
The reaction of CO with O2 over Pd catalysts forming CO2 is one of the most studied reactions in catalysis, both due to its practical relevance and due to its role in studies of structure-function correlations and (surface) oxide formation45,46. Therefore, we chose it as our model reaction with the aim to enable QMS measurements of the CO2 formed on a single Pd nanoparticle. We have developed a nanofluidic reactor connected to U-shape microfluidic in- and a straight outlet channel(s), which are connected to a macroscopic reactant inlet system operated by mass flow controllers at 3 bar, and to a QMS mounted on a UHV chamber with 1 ⋅ 10−10 mbar base pressure (Fig. 1a and Supplementary Fig. 1). This in- and outlet system is connected to a nanofluidic catalyst bed with 30 μm × 10 μm × 200 nm(L × W × H) dimensions via a nanofluidic inlet channel (505 μm × 10 μm × 200 nm) and an outlet channel with dimensions 100 μm × 500 nm × 200 nm (Fig. 1b, c). This design ensures that the catalyst is operated in the well-mixed regime, where concentration gradients due to reactant conversion are effectively eliminated35. Furthermore, we note that the gas flow in such a system is in the slip flow and transitional flow regimes, with the Knudsen number increasing in the mean flow direction throughout the nanofluidic reactor35. Consequently, momentum, heat and mass transfer boundary layers surrounding reacting particles are thinner than in the continuum regime, and the mass diffusivities approach the Knudsen diffusivity. In fact, nanofluidic reactors therefore constitute effective “model pores” since the aforementioned effects all are present in technically widely used porous catalyst systems36. Using a resistive heater, the nanoreactor zone can be heated up to 450 °C36. For this study, we have fabricated four nanoreactor chips with identical gas in- and outlet systems, as well as catalyst beds. Using electron-beam lithography (EBL) nanofabrication47, they were decorated with (regular arrays of) n = 1000, 10, 1 or 0 Pd particles with diameter d = 59.4 ± 3.7 nm and height h = 23.5 ± 1.6 nm, as SEM image analysis reveals, physical vapor deposited (PVD) onto a 8 nm thick SiO2 support layer that separates them from a chemically inert plasmonic Au nanoparticles previously PVD-grown through the same EBL mask with d = 97.6 ± 6.6 nm and h = 35.3 ± 2.3 nm (Fig. 1c–g and Supplementary Fig. 2 and Methods section on nanofabrication of the nanoreactor chip36,48. This corresponds to an active Pd surface area A ≈ (0.0072 ± 0.00086) μm2 = (7200 ± 860) nm2 per particle. We chose this particular hybrid nanoparticle design, where the Au element serves as a plasmonic light scatterer (Pd is optically dark49), to enable the verification of the presence of the anticipated number of particles in the enclosed nanoreactors using dark-field scattering microscopy (Fig. 1d–g).

a Schematic of the micro- and nanofabricated reactor chip. It is comprised of a U-shaped microfluidic in- and a simple straight outlet system that connects to the model catalyst bed (the nanofluidic reactor with the reaction zone) on either side, as well as to the high-pressure gas handling system and the QMS, respectively. The cross-sectional dimensions of the microfluidic channels are also shown. b Schematic depiction of the 30 μm long well-mixed reaction zone (the model catalyst bed that contains the nanoparticle(s)) with cross sectional dimensions 10 μm × 200 nm, as well as its connections to the microfluidic in- and outlet system via a smaller microfluidic inlet channel (also with cross sectional dimensions 10 μm × 200 nm) and via a nanofluidic outlet “capillary” with cross sectional dimensions 500 nm × 200 nm. Note that the schematics are not drawn to scale. c Dark-field scattering microscopy image of a nanofluidic catalyst bed containing 1000 nanoparticles arranged in a regular array in the reaction zone. The inset depicts a side-view scanning electron microscopy (SEM) image of the used Au/SiO2/Pd hybrid nanostructures. Scale bar 100 nm. The catalytically inert Au element enables single-particle dark-field scattering (DF) imaging to confirm the presence of the correct number of catalyst particles inside the sealed nanofluidic reactor, without itself participating in the reaction36. d Schematic depiction and zoomed-in DF image of the reaction zone of the nanofluidic catalyst bed used in our experiments, with a regular array of n = 1000 nanoparticles that become distinctly visible in the DF image. Also shown are two top-view SEM images of a similar array of nanoparticles, as well as a side-view of a single nanoparticle, prepared on an open surface to facilitate SEM imaging. Scale bar 100 nm. The inset image below “SEM” includes a scale bar for reference. e Schematic depiction and zoomed-in DF image of the reaction zone of the catalyst bed containing n = 10 Pd nanoparticles used in our experiments. f Schematic depiction and zoomed-in DF image of the reaction zone of the catalyst bed containing a single (n = 1) Pd nanoparticle used in our experiments. g Schematic depiction and zoomed-in DF image of the reaction zone of the empty catalyst bed without Pd nanoparticles (n = 0), used in our experiments as a negative control.
CO oxidation on Pd model catalysts in nanofluidic reactors
To prepare these systems for experiments in reaction environment, we first exposed them to a conditioning sequence, followed by a full CO oxidation sequence at 280 °C (see Methods section on sample mounting and pre-treatment and Supplementary Figs. 3–6). Subsequently, we initiated the experiment sequence consisting of 15 min CO/O2 mixture pulses at a constant 6% reactant concentration and starting with 6% percent CO, separated by 15 min in Ar. Such sequences were executed from 280 to 450 °C in 20 °C steps for the n = 1000 sample and 10 °C steps for the n = 10 and 1 samples, to sweep the entire \(0\le {\alpha }_{{{{\rm{CO}}}}}={P}_{{{{\rm{CO}}}}}/({P}_{{{{{\rm{O}}}}}_{2}}+{P}_{{{{\rm{CO}}}}})\le 1\) range in steps of 0.05, where αCO is the relative CO concentration in the gas mixture. (Fig. 2a–c). At each new temperature, a single 30 min pulse of 4% O2 and 2% CO in Ar carrier gas, followed by a 15 min pure Ar pulse, was applied to reset the state of the catalyst.

CO and O2 pulse sequence applied at a reactor temperature of (a) T = 450 ∘C, (b) T = 340 ∘C, (c) T = 280 ∘C. Note that we start all experiments with a pure 6% CO pulse in Ar carrier gas and subsequently decrease the relative CO concentration and increase the relative O2 concentration, such that \(0\le {\alpha }_{{{{\rm{CO}}}}}={P}_{{{{\rm{CO}}}}}/({P}_{{{{{\rm{O}}}}}_{2}}+{P}_{{{{\rm{CO}}}}})\le 1\), while keeping the total reactant concentration constant at 6%. Corresponding baseline-adjusted (BA – see Methods section on preprocessing for explanation of the BA-procedure) CO2 counts measured by the QMS for 1000 Pd nanoparticles at (d) T = 450 ∘C, (e) T = 340 ∘C and (f) T = 280 ∘C. We note distinct responses at all three temperatures with the expected decrease in reaction rate for lower T. The “transient” overshoots observed for small αCO values are the consequence of the mass flow controller “dial-in” to the correct flow rate. (g–i) Corresponding CO2 counts measured by the QMS for the 10 Pd nanoparticle sample. While at T = 450 ∘C a relatively clear CO2 signal is still obtained, it becomes increasingly weaker at the lower temperatures. j Mean value of the measured CO2 QMS counts for each reactant pulse plotted vs. the corresponding αCO value for both the n = 1000 and the n = 10 Pd nanoparticle samples and obtained at T = 450 ∘C. Shaded area depicts the standard deviation of measured BA-counts across each respective pulse. We note a maximum in the CO2 formation rate at \({\alpha }_{{{{\rm{CO}}}}}^{\max }=0.65\) for both n = 1000 and 10, as well as a significantly larger deviation of the derived counts for n = 10. k Same as (j) but for T = 340 ∘C. We note a shift of \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) to lower values for both samples due to increasing CO poisoning and a broadening/flattening of the overall trend for n = 10, which is the consequence of the system approaching the detection limit of the QMS and the consequent significant uncertainty in the derived counts. l Same as (j) and (k) but for T = 280 ∘C. We note an even further reduction of \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) and further uncertainty increase for n = 10. Source data are provided as a Source Data file.
Focusing first on the highest reaction temperature of 450 °C and n = 1000 (total active surface area of A ≈ (7200 ± 860) nm2 ⋅ 1000 = (7.2 ± 0.86) μm2), we observe a QMS response distinctly above the noise floor for all αCO ∈ (0, 1) values (Fig. 2d). Furthermore, starting from αCO = 1, we observe the characteristic reaction rate increase as more O2 is added until the maximum rate is reached at \({\alpha }_{{{{\rm{CO}}}}}^{\max }=0.65\). This is very close to the stoichiometric αCO = 0.66 (the small difference is the consequence of the 0.05 αCO steps), which indicates that CO poisoning is very mild at this temperature and that mass transport gradients are negligible, as expected for a well-mixed catalyst bed. Upon decreasing αCO beyond \({\alpha }_{{{{\rm{CO}}}}}^{\max }\), the reaction rate decreases in an essentially linear fashion.
Decreasing the temperature to 340 °C induces both a global decrease in reaction rate and a shift of \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) to a lower value of 0.5, due to stronger CO poisoning (Fig. 2e). This trend continues upon temperature reduction to 280 °C (Fig. 2f), with a QMS signal still distinctly above the noise floor and \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) reduced to 0.3. These measurements thus corroborate our experimental setup and approach since they deliver results in agreement with the well-established understanding of the CO oxidation over Pd catalysts45,46. Hence, they constitute a relevant baseline as we further reduce the catalyst surface area towards a single Pd nanoparticle.
As the first step, we repeated the experiments for an identical nanoreactor but with n = 10, which corresponds to A ≈ (7200 ± 860) nm2 ⋅ 10 = (0.072 ± 0.0086) μm2 (Fig. 2g–i). We find that at 450 °C, CO2 pulses still are resolved and that the counts is ≈ 2 orders smaller compared to n = 1000. Importantly, however, we also see that the CO2 signal induced by the reactant pulses approaches the noise floor. Accordingly, reducing the temperature makes discerning the CO2 produced increasingly difficult. This becomes clear when extracting the CO2 counts for each αCO pulse and directly comparing the CO2 counts vs. the αCO trend for the n = 1000 and 10 samples. At T = 450 °C, the trends obtained are qualitatively very similar and thus corroborate that the catalyst is operated at identical conditions (Fig. 2j). It is also visible that the counts vs. αCO curve is slightly wider and flatter, and exhibits a significantly larger standard deviation (shaded area) for n = 10, in particular in the low reaction rate regimes at small and high αCO values. Reducing temperature further amplifies this effect as the signal approaches the noise floor for n = 10, with counts in the 10–40 range at T = 280 °C (Fig. 2k, l). Consequently, it becomes increasingly difficult to extract the CO2 signal, and we are approaching the limit of detection from a conventional data analysis perspective, where simply the number of counts is extracted from the QMS signal.
In principle, various strategies to address this issue exist, e.g., digital filtering50, Fourier transforms51,52, wavelet transforms53, etc., or statistical methods, like principal component analysis (PCA)54, among others. When applied correctly, they enable the extraction of the desired signal from the accompanying noise and enhance the visibility of the underlying patterns in the data. However, these well-established denoising techniques have inherent limitations in the context of denoising QMS signals. Specifically, digital filtering can inadvertently remove crucial signals if they overlap with noise frequencies55, Fourier and wavelet transforms may falter if the signal doesn’t meet their assumptions about periodicity or localization56 and PCA, which assumes the primary variance in data is due to the signal, might not be effective if noise has high variance or the signal is confined to a tight range57. Given these challenges, a denoising auto-encoder (DAE), i.e., an artificial neural network, offers a promising solution since it can be trained to discern the structure of complex noisy data and reconstruct the underlying signal58 (see Supplementary Note 3 and Supplementary Table 1). In practice, this is achieved by introducing examples of simulated signals with experimentally measured noise and training the network to reconstruct the (original) simulated signal. By also constraining the autoencoder’s latent space to have a step function distribution, we introduce a prior, which enables denoising well below the signal-to-noise ratio (SNR) of 1 and ensures robustness in the final representations59.
A constrained DAE to improve the QMS limit of detection
We applied the DAE to our QMS experiments by using experimentally measured QMS readout consisting of intrinsic measurement noise and noise induced by the nanoreactor setup (e.g., fluctuations in CO2 concentration stemming from impurities in the used gases, small fluctuations of the mass flow controller and/or tiny leaks), combined with an underlying true CO2 signal stemming from the catalytic reaction on the Pd nanoparticle(s), as input (Fig. 3). This combined signal is then compressed through encoder Eθ to form a latent space inside the bottleneck, which is constrained through a consistency loss to form a step function distribution. This compressed representation is then upsampled in decoder Dθ to form the reconstructed true signal, i.e., the QMS readout with a deconvolved underlying true CO2 signal. In this design, the DAE can learn complex non-linear relationships between the noise and the true signal, enabling it to handle the kind of noise-signal interactions that might confound the other noise-reducing methods mentioned above. For a comparison between the DAE and other denoising techniques applied to our QMS data, see Supplementary Note 4 and Supplementary Figs. 7–9.

The underlying signal that we want to access in our experiments, i.e., the CO2 production rate step functions as a function of time, is corrupted by both correlated and uncorrelated noise in the experimentally measured readout from the QMS. Therefore, the experimental QMS readout is input to a denoising autoencoder, which transforms the input into a minimally dimensioned representation of the underlying signal and then reconstructs it sans corruption, which means, it reconstructs the original signal, rather then the QMS readout that also contains different types of noise. The DAE is trained with a standard L2 norm between reconstructed and underlying signal, and the latent space is constrained by an L1 norm and a consistency loss, which forces the latent space to take on the form of the underlying signal.
To evaluate the performance of the DAE, we compare the measured baseline-adjusted (BA) QMS CO2 counts (see Methods section on preprocessing for explanation of the BA-procedure) as function of αCO for n = 1000 and 10, for temperatures between 280 and 450 °C, as well as the DAE-denoised CO2 counts for n = 10 (Fig. 4). Focusing first on n = 1000 at 450 °C, the catalyst exhibits the highest reaction rate at \({\alpha }_{{{{\rm{CO}}}}}^{\max }=0.65\) and all CO2 pulses are clearly discernible (Fig. 4a). Executing identical analysis for n = 10 at 450 °C reveals individual CO2 pulses and \({\alpha }_{{{{\rm{CO}}}}}^{\max }\approx 0.6\) (Fig. 4b). Passing the n = 10 QMS data obtained at 450 °C through the DAE reveals that it delivers more distinct and systematically in/decreasing CO2 pulses, with a maximum rate at \({\alpha }_{{{{\rm{CO}}}}}^{\max }=0.65\), in excellent agreement with n = 1000 (Fig. 4c). Repeating the same analysis for 280 °C reveals the anticipated reduction of the overall rate for both samples and a distinct shift of \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) to \({\alpha }_{{{{\rm{CO}}}}}^{\max }\approx 0.2\), due to severe CO poisoning (Fig. 4d–f). Importantly, for n = 10, it is now clear that the standard analysis is approaching the limit of detection where the CO2 pulses are stochastic, whereas the DAE-denoised data still exhibits the same clear trend as seen for n = 1000 with the standard analysis.

a Baseline-adjusted (BA – see Methods section on preprocessing for explanation) raw QMS counts for CO2 (gray line) together with the mean CO2 signal (blue line – obtained as the average measured BA-count across each pulse) for n = 1000 Pd nanoparticles across the entire αCO range, αCO ∈ (0, 1), and at 450 ∘C. b BA-QMS counts for CO2 (gray line) together with the mean CO2 signal (green line) for n = 10 Pd nanoparticles across the entire αCO range and at 450 ∘C. c BA-QMS counts for CO2 (gray line) together with the CO2 signal denoised by the DAE (orange line) for n = 10 across the entire αCO range, αCO ∈ (0, 1), and at 450 ∘C. (d–f) Same as (a–c) but at 280 ∘C. g BA-mean CO2 counts for αCO sweeps at reactor temperatures ranging from 280 to 450 ∘C in 20 ∘C steps for n = 1000. Red dots indicate the αCO value corresponding to the highest reaction rate, \({\alpha }_{{{{\rm{CO}}}}}^{\max }\). h Same as (g) but for n = 10, using 10 ∘C temperature steps. i Same as (h) but with the QMS signal denoised by the DAE. j \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) values extracted from (g) for all temperatures for n = 1000, plotted as a function of αCO. We note the constant stoichiometric \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) value of 0.65 down to T = 360 ∘C. Below this temperature, we find a systematic shift to lower \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) values, as a consequence of increasing CO poisoning. k Same as (j) but for n = 10. We note the qualitatively similar trend compared to n = 1000, but also the significantly higher spread in the data points. l Same as (k) but based on the DAE-denoised QMS signal in (i). Clearly, the uncertainty in \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) is significantly reduced and the T-dependent trend of \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) for both n = 1000 and 10 is now very similar. The small discrepancies are discussed in the text. Source data are provided as a Source Data file.
As the next step, we extract the mean CO2 counts for each αCO pulse along the αCO sweep for n = 1000, obtained for temperature steps of 20 °C from 280 to 450 °C (Fig. 4g). This reveals a constant and stoichiometric \({\alpha }_{{{{\rm{CO}}}}}^{\max }=0.65\) down to 380 °C, below which \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) systematically shifts to smaller values due to increasing CO poisoning, and reaches \({\alpha }_{{{{\rm{CO}}}}}^{\max }=0.25\) at 280 °C. Furthermore, it reveals a decrease in reaction rate from 5240 CO2 counts at \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) at 450 °C, to 1700 CO2 counts at \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) at 280 °C. Plotting the same data for n = 10 obtained for 10 °C temperature steps, and comparing the standard analysis (Fig. 4h) with the DAE-based analysis (Fig. 4i) reveals qualitatively very similar behavior. However, crucially, SNR is very poor for the standard analysis to the point where the underlying chemical dynamics become obscured by stochastic shifts in, e.g., \({\alpha }_{{{{\rm{CO}}}}}^{\max }\). This is different for the DAE-based data, which reproduces the T-dependent \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) trend found for the n = 1000 sample very clearly (Fig. 4j–l). The situation is similar when comparing the absolute BA – CO2 counts at \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) at 450 °C. For n = 1000, we count 5240 CO2 molecules, whereas we count 90 and 65 for n = 10 for the standard and DAE-based analysis, respectively. The DAE-based value of 65 is indeed proportional to a 100 × decrease in catalytic surface area (compared to n = 1000), within a relative error of roughly 10%. Thus, the measured raw value of 90 counts likely overestimates the true CO2 production rate due to residual noise or background signal, highlighting the DAE’s ability to accurately correct for that. The results demonstrate that the DAE effectively reconstructs the catalysts’ CO2 production rate and, e.g., the temperature-dependent poisoning state of the system, even when dealing with considerably noisy data and the QMS operated very close to its limit of detection.
Resolving reaction products formed on Pd NPs with a DAE
We put the DAE to the test by further reducing the amount of catalyst by a factor 10, down to a single Pd nanoparticle with a surface area of ≈ 7200 nm2. We used a chip with n = 1 (cf. Fig. 1f) and an empty chip with n = 0 as the control (cf. Fig. 1g). Clearly, even at the highest temperature of 450 °C, the αCO sweeps for n =0 and 1 look very similar using the standard analysis (Fig. 5a, b). They are characterized by small stochastic CO2 pulses, indicating a certain level of background CO2 in the reactant gas mixture, as well as the signal stemming from the CO2 produced by the catalyst particle drowning in background noise. This is further corroborated by extracting the BA-CO2 counts and plotting them vs. αCO for both n = 1 (7 identical sweeps) and 0 (5 identical sweeps), which reveals that no clear activity trend as a function of αCO is resolved for n = 1, despite a generally slightly higher number of counts compared to n = 0 (Fig. 5c and Supplementary Figs. 6, 10).

a Baseline-adjusted (BA) raw QMS counts for CO2 (gray line) together with the mean CO2 signal (green line – obtained as average measured BA-count across each pulse) for n = 1 Pd nanoparticle, measured across the entire αCO range, αCO ∈ (0, 1), and at 450 ∘C. b Same as (a) but for an empty nanochannel, i.e., n = 0. Note the very similar and stochastic appearance of apparent CO2 pulses both for n = 1 and 0 when analyzed in the standard way. c Mean BA-CO2 counts extracted for each αCO pulse based on the standard analysis across the entire αCO range, αCO ∈ (0, 1), and at 450 ∘C, for both n = 0 (black lines) and 1 (green lines). For n = 1 we executed 7 consecutive αCO sweeps, and for n = 0 we executed 5 consecutive αCO sweeps. As key observation, we note high reproducibility of the αCO sweeps for both n = 0 and 1, as well as that no clear activity trend as a function of αCO is resolved for the n = 1 sample, despite a generally slightly higher number of counts compared the n = 0 reference. d Same as (a) but for DAE-denoised BA-QMS CO2 counts (purple line). Note the significantly smaller but at the same time non-stochastic CO2 pulses resolved by the DAE compared to the standard analysis. e Same as (d) but for an empty nanochannel, i.e., n = 0. Note that using the DAE, a flat baseline at zero BA-counts is obtained for the empty nanochannel. f Same as (c) but for DAE-denoised data, where distinct reaction rate maxima are resolved for n = 1 (purple curves), with \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) ranging between 0.65 and 0.6 for temperatures between 450 and 410 ∘C (inset), in good agreement with the earlier results for n = 10 and 1000. For lower temperatures, as well as for n = 0 (black lines representing multiple sweeps at 450 ∘C), the DAE outputs counts < 1, which is physically unreasonable and thus defined as the limit of detection. g \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) values for n = 1000 (blue), 10 DAE-denoised (orange) and 1 DAE-denoised (purple) for 400 ∘C≤ T ≤ 450 ∘C, i.e., the range for which the n = 1 DAE-signal is above the limit of detection. h CO2 counts normalized by the number of particles on the respective sample, obtained at \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) for n = 1000 (blue), n = 10 DAE-denoised (orange) and 1 DAE-denoised (purple) for 400 ∘C ≤ T ≤ 450 ∘C. Source data are provided as a Source Data file.
Applying the DAE first to the αCO sweep at 450 °C for n = 1 reveals a different picture (Fig. 5d). Rather than stochastic CO2 pulses, a clear trend of in- and decreasing CO2 counts along the αCO sweep is recovered, which again exhibits a maximum at the stoichiometric \({\alpha }_{{{{\rm{CO}}}}}^{\max }=0.65\). This is in very good agreement with the corresponding experiments for n = 10 and 1000. Applying the DAE to the αCO sweep at 450 °C for the n = 0 control outputs a close to completely flat baseline at zero BA-counts, corroborating that the DAE is able to eliminate the noise contaminating the raw QMS signal (Fig. 5e).
Encouraged, we apply the DAE to αCO sweeps measured at temperatures 330 to 450 °C for n = 1 and plot the extracted CO2 counts vs. αCO (Fig. 5f). Remarkably, we clearly resolve the anticipated increase of the reaction rate to an \({\alpha }_{{{{\rm{CO}}}}}^{\max }=0.65\) for decreasing αCO, and the subsequent decrease of the reaction rate upon decreasing αCO. Furthermore, the extracted number of CO2 counts ≈ 6 is ≈ 10 times lower than for n = 10 at 45 °C, which is in excellent agreement with a reduction of the active surface area by a factor of 10 from n = 10 to 1. Finally, upon reduction of temperature, we see an indication of a shift of \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) to lower values at 410 °C, before the DAE-signal also drops below the limit of detection of a single count. Similar analysis of the n = 0 control at 450 °C consistently delivers CO2 counts very close to or significantly below the limit of detection of a single count and thus confirms the ability of the DAE to resolve CO2 produced by the single Pd nanoparticle (Fig. 5f). As a last point, we note that the low absolute counts observed for single nanoparticles (c.f. Pd n = 1 in Fig. 5f) raise the question of potential error in the activity analysis in this regime of very few counts. While it is true that low count numbers can, in principle, lead to larger relative uncertainties, the DAE’s denoising capabilities mitigate this issue. Specifically, the DAE is trained to distinguish genuine signals from noise, even when the signal is very weak. Therefore, while the absolute number of counts might be low, the proportion of those counts representing actual CO2 production is very high after DAE processing compared to the raw data and thus the counts predicted by the DAE are indeed significant. Furthermore, the consistency of the DAE-derived trends across multiple independent measurements (c.f. Fig. 5) corroborates the reliability of the activity trends obtained by the DAE, even for single nanoparticles.
We put these results into perspective by comparing them to the data obtained for n = 10 and 1000. Specifically, we plot the \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) values for n = 1000, 10 DAE-denoised and 1 DAE-denoised for 400 °C ≤ T ≤ 450 °C, i.e., the range for which the n = 1 DAE-signal is above the limit of detection (Fig. 5g). These values are very similar for all three samples and very close to the stoichiometric value of \({\alpha }_{{{{\rm{CO}}}}}^{\max }=0.66\). This is important because it confirms that the catalyst experiences identical reaction conditions in all three implementations in terms of the number of particles. We also compare the CO2 counts obtained at \({\alpha }_{{{{\rm{CO}}}}}^{\max }\) for 400 °C ≤ T ≤ 450 °C normalized by the number of particles on the respective sample, i.e., n = 1000, 10 and 1 (Fig. 5h). This reveals particle-number-normalized CO2 count values between 4 and 6 and thus corroborates direct scaling of extracted QMS counts with catalyst surface area, within the expected level of uncertainty that is imposed, e.g., by slightly different particle dimensions on each of the three chips.
To ensure that the CO2 signal observed in the catalytic experiments originates exclusively from surface reactions on Pd nanoparticles, we conducted a control experiment using an identical nanofluidic chip lacking any catalytically active material. Pulses of CO2 diluted in Ar were flushed through the chip under identical conditions (450 °C, 2 bar, 20 mL min−1 total flow), and the resulting QMS signal was analyzed. As detailed in Supplementary Fig. 17a, b, no evidence of spurious CO2 formation or retention effects was observed. Furthermore, the DAE successfully retrieved the shape and timing of the low-intensity CO2 pulses even when the raw signal approached the noise floor, confirming the robustness of the signal processing method and excluding non-catalytic sources of CO2 response.
DAE for ethylene hydrogenation with a less sensitive QMS
To demonstrate the versatility of the DAE presented in this work, as well as to test it from a different perspective and using a different catalytic reaction, we investigated ethylene hydrogenation on 1000 Pd nanoparticles using a QMS with lower intrinsic sensitivity. This experiment was designed to illustrate that our system is not limited to CO oxidation and not only can reduce the amount of catalyst surface area required for online QMS measurements but also can be used to increase the sensitivity of lower grade QMS instrument. Therefore, we exchanged the high-sensitivity Hiden HAL/3F PIC QMS used in the CO oxidation experiments by a significantly less sensitive Pfeiffer Prisma QME200. Furthermore, as in the CO oxidation experiments, we used a pulsed gas sequence comprised of alternating pulses of pure Ar and C2H4 mixed with H2 in different ratios in Ar at a total constant reactant concentration of 1% and 4 bar inlet pressure (Fig. 6a). This sequence was designed to transition from pure C2H4 to pure H2, and back to pure C2H4, while monitoring the formed ethane (C2H6) with the QMS. The experiment was conducted at temperatures of 85, 90, 120, 160, and 170 °C, respectively. Analyzing the raw normalized C2H6 ion current measured by the QMS alongside the filtered signal derived using the DAE for three representative temperatures reveals a distinct QMS response at the highest temperature of 170 °C that quickly drowns in noise when the reaction temperature is reduced (Fig. 6b, see Supplementary Fig. 16 for all measured temperatures). Accordingly, it again showcases the ability of the DAE to extract the weak QMS signals from noise in this regime, as the DAE predicted C2H6 QMS response exhibits clear pulses even at the lowest temperature of 85 °C.

a Mixed C2H4 and H2 pulses with systematically varied concentrations in Ar carrier gas applied to the 1000 Pd nanoparticle catalyst bed at different temperatures. b Baseline-adjusted (BA – see Methods section on preprocessing for explanation) normalized C2H6 reaction product counts measured by QMS at m/z = 30 (gray lines), together with the signal denoised by the DAE (colored lines) for 85, 120, and 170 ∘C catalyst bed temperatures. The colors refer to the corresponding temperatures shown in panel (c). c, d BA-adjusted C2H6 counts as a function of the reactant mixing parameter \({\alpha }_{{{{{\rm{C}}}}}_{2}{{{{\rm{H}}}}}_{4}}={P}_{{{{{\rm{C}}}}}_{2}{{{{\rm{H}}}}}_{4}}/({P}_{{{{{\rm{C}}}}}_{2}{{{{\rm{H}}}}}_{4}}+{P}_{{{{{\rm{H}}}}}_{2}})\), calculated for every step of the pulse sequence shown in (a). The arrows indicate the direction of the sequence: in (c) \({\alpha }_{{{{{\rm{C}}}}}_{2}{{{{\rm{H}}}}}_{4}}\) goes from 1 to 0 in steps of 0.1, in (d) \({\alpha }_{{{{{\rm{C}}}}}_{2}{{{{\rm{H}}}}}_{4}}\) goes from 0 to 1 in steps of 0.1. The colors in (d) match the temperatures reported in (c). Source data are provided as a Source Data file.
To further analyze the obtained results, and in analogy to the CO oxidation experiments, we plot the averaged ion counts for each step of the pulse sequence predicted by the DAE as a function of the mixing parameter \({\alpha }_{{{{{\rm{C}}}}}_{2}{{{{\rm{H}}}}}_{4}}={P}_{{{{{\rm{C}}}}}_{2}{{{{\rm{H}}}}}_{4}}/({P}_{{{{{\rm{C}}}}}_{2}{{{{\rm{H}}}}}_{4}}+{P}_{{{{{\rm{H}}}}}_{2}})\) for the down-sweep (Fig. 6c) and up-sweep (Fig. 6d), respectively. In the down-sweep, as the concentration of C2H4 decreases, the C2H6 production initially peaks and then decreases, indicative of first-order kinetics with respect to C2H4, followed by a regime of negative-order kinetics. Conversely, in the up-sweep, the system exhibits first-order kinetics relative to H2, followed by a zero-order plateau, and then transitions into negative-order kinetics. This behavior is attributed primarily to the competitive adsorption between ethylene and hydrogen on the Pd catalyst surface. During the up-sweep, H atoms accumulate more rapidly than ethylene can compete for adsorption sites, resulting in a surface saturated with H and thus a zero-order kinetic regime with respect to H260,61. In contrast, starting with a surface saturated with C2H4 during the down-sweep hinders H adsorption once introduced, preventing the establishment of the zero-order regime62. These findings align with the Horiuti-Polanyi mechanism, where surface coverage and competitive adsorption play crucial roles63,64. Overall, these results demonstrate that the DAE in combination with nanofluidic reactors enables the extraction of detailed kinetic information also from a much slower hydrogenation reaction, and that at lower overall reactant concentration and with a less sensitive QMS than for the CO oxidation experiments above, using a small catalyst bed of 1000 Pd nanoparticles.
link





.jpg)