Roboticized AI-assisted microfluidic photocatalytic synthesis and screening up to 10,000 reactions per day

Ultra-fast photocatalytic synthesis and characterization
We designed and constructed a new type of microfluidic photocatalytic microreactor using the LCW technique to introduce high-intensity laser light into the microfluidic photocatalytic reaction channel to significantly increase the speed of photocatalytic reaction (Fig. 1A). We used four 450-nm lasers as light sources and coupled a total of ca. 4.6 W of light into the reaction area through four optical fibers. The light transmitted by multiple total reflections in the 5-cm-long reaction channel based on the LCW principle, resulting in a laser luminous flux over 3.5 × 104 mW/cm2 through the channel cross section within a channel length range of 3 cm (Fig. 1B). Such a light irradiation intensity is ca. 10,000 times more than those in conventional batch photocatalytic systems28,29. In the microfluidic LCW microreactor, the laser light was reflected multiple times in the photocatalytic microreactor, resulting in a more uniform high-intensity light distribution in the 3-cm-length middle part of the 530-μm-diameter reaction channel, which was 15 times more than the ca. 2 mm irradiation depth in conventional batch photocatalytic systems. However, we observed that the use of the high-intensity laser irradiation caused a significant increase in temperature in the capillary reactor, resulting in an increase in side reactions and a decrease in reaction yield. To address this problem, we designed the microfluidic LCW photocatalytic microreactor as a cannula configuration with an inner reaction capillary and an outer capillary with the circulating cooling solution flowing through to control the temperature of the reaction channel, for instance, 25 ± 2 °C.
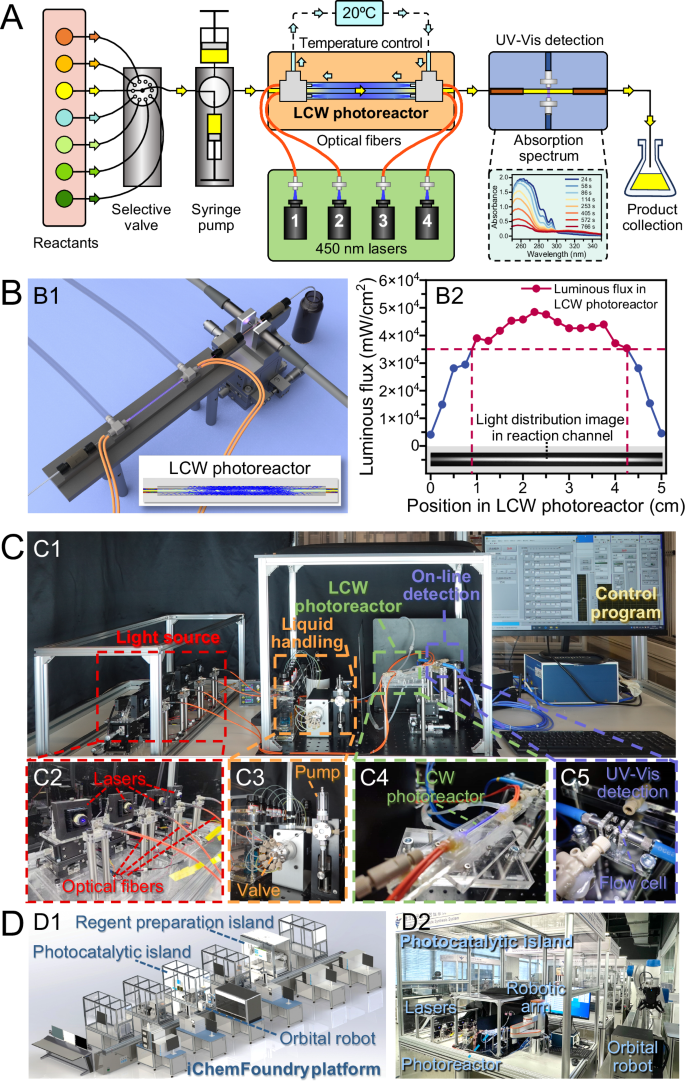
A Schematic diagram of the system flow manifold. B Structure and light distribution of the LCW photocatalytic microreactor. (B1) Schematic diagram of the LCW photocatalytic microreactor and online UV-Vis absorption spectroscopic detection device; (B2) Cross-sectional light intensity distribution in the reaction channel of the LCW photocatalytic microreactor obtained using TracePro software. C Photographs of the system. (C1) Overview of the system. (C2) Light source module, consisting of four 450-nm lasers and four optical fibers. (C3) Liquid handling module with a 10-port selective valve and a syringe pump. (C4) LCW photocatalytic microreactor. (C5) Online UV-Vis absorption spectroscopic detection module. D Schematic diagram (D1) and photograph (D2) of the iChemFoundry platform.
We applied the LCW photocatalytic microreactor to a typical organic photocatalytic synthesis, photocatalytic [2 + 2] cycloaddition reaction30, which has gained extensive attention because cyclobutane obtained through this reaction has been frequently found in a variety of natural products with biological activity31,32,33. Using the LCW photocatalytic microreactor, the photocatalytic [2 + 2] cycloaddition reaction of substrate S-1 could be completely converted with a residence time of only 3.3 s in the microreactor channel, while the reaction yield and diastereomeric ratio (d.r.) were comparable to those of conventional batch photocatalytic systems. As a comparison, the time consumption of this photocatalytic [2 + 2] cycloaddition reaction in a conventional batch photocatalytic reactor was up to 4 h with the same photocatalyst and substrate species, concentration and photocatalyst ratio conditions. Our LCW photocatalytic microreactor could shorten the reaction time by 4300 times with the aid of ultra-high light intensity, uniform and long-length light irradiation, stable reactor temperature control and microfluidic scale effect. To the best of our knowledge, this is the first report of shortening a photocatalytic reaction from several hours to a few seconds, reaching the fastest photocatalytic reaction speed reported in the literature.
In addition to high synthetic reaction speeds, the characterization speed of the reaction products or reactants needs to match the reaction speed to achieve real high-throughput screening. Currently, the routine analysis of organic synthesis products is usually performed by chromatography, mass spectrometry and nuclear magnetic resonance spectroscopy (NMR), most of which are difficult to achieve high-throughput and rapid analysis at the second level34. Spectroscopic analysis has the advantages of non-destructive, rapid and easy to implement online detection. Although its weaknesses in qualitative analysis capability and selectivity limit the application for accurate analysis of reaction products, it is well suited in many systems for online monitoring the changes of unsaturated groups in the synthetic reaction process to provide decisive data for the initial high-throughput screening. Before and after the cycloaddition reaction performed in this work, there was a significant change in the UV spectrum of the reaction solution (caused by the consumption of the substrate) in the region of 280 nm–320 nm, thus the reaction solution could be online detected by coupling a capillary flow cell to the LCW photocatalytic microreactor and using a UV-Vis spectrometer with a detection response time of 0.1 s. We also examined the reliability of the UV-Vis detection method in reaction monitoring using the routine characterization methods for organic synthesis reactions—gas chromatography (GC) and NMR.
High-throughput photocatalytic synthesis and screening
In addition to the ability of rapid synthesis and online characterization, a practical high-throughput system must have the ability to automatically change both continuous variables (e.g., reaction temperature, time, pressure, light intensity, and reactant concentration, etc.) and discrete variables (e.g., substrate, photocatalyst and solvent species, etc.) for performing meaningful high-throughput screening of synthesis conditions. In many cases, much attentions are usually paid to achieving rapid organic synthesis reactions under specific conditions, the ability to achieve rapid screening of a large number of different conditions is frequently overlooked. In the present system, we used a liquid handling module consisted of a selective valve and a syringe pump to achieve automated multistep liquid handling operations, including rapid introducing, changing, mixing and driving of different reaction solutions with adjustable flow rates. We uniquely used a combination of a 1-mL and a 5-mL syringes to perform multi-step reciprocal aspirating-dispensing operations for rapidly achieving automated mixing of mL-scale of reactants (substrate, photocatalyst and solvent) within 2 min. We designed a LabVIEW-based program to control the operation of the whole modules of the system (Fig. 1C), allowing the on-demand automated changing of photocatalyst and substrate species, concentration, photocatalyst ratio, laser light intensity and flow rate to achieve the fully-automated operations of the large-scale screening of reaction conditions (>10,000 reactions) without the need of any manual operation and intervention. This photocatalytic synthesis and screening system also served as one of multiple functional islands of the iChemFoundry (IC) platform. The IC platform is a large-scale automated platform for molecular manufacturing which consisted of various functional islands for realizing a series of automated operations of chemical synthesis from reactant preparation, pre-treatment, chemical synthesis reactions, post-treatment, characterization and data analysis. By combining the functional islands of the photocatalytic synthesis and screening system, the reactant preparation system, an orbital robot and an on-island robotic arm of the IC platform, complete automation of all operations could be achieved, including solid reagent weighing, liquid reagent metering, preparation of reagent stock solutions, transferring and aspirating of reagent stocks, preparation and introduction of reactant solutions, on-line flow photocatalytic reactions, UV spectroscopic reaction monitoring, as well as cyclic screening of different conditions (Fig. 1D).
With the present system, we performed a comprehensive screening for the photocatalytic [2 + 2] cycloaddition reaction with a total number of up to 12,000 conditions, including two discrete variables of the photocatalyst and substrate species and four continuous variables as the laser light intensity, concentration, flow rate, and photocatalyst ratios (Fig. 2). Each of the six variables had 4–6 levels or species, resulting in a total orthogonal combination number of 12,000 screening conditions (i.e., photocatalytic synthesis and analysis experiments). In our system, the average time required to obtain the data for each photocatalytic [2 + 2] cycloaddition reaction was only 32 s, which enabled the system to reach a screening throughput of 2600 conditions per day.
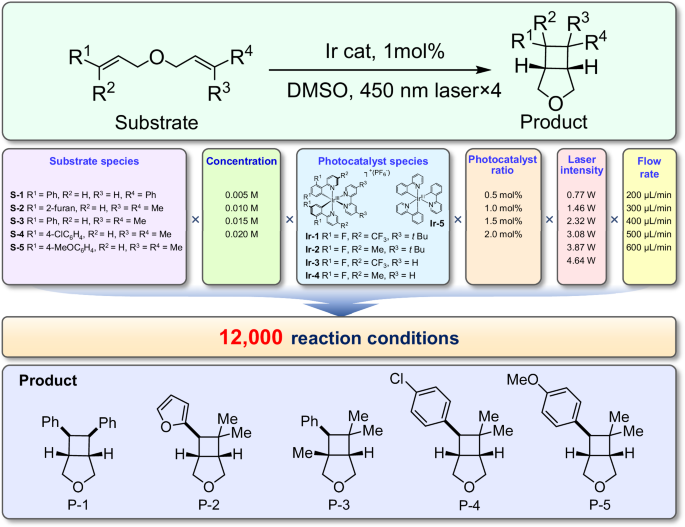
List of variables and conditions screened in the photocatalytic [2 + 2] cycloaddition reaction.
AI-assisted ultra-high-throughput photocatalytic synthesis and screening
Although the above throughput of 2600 conditions per day with a single flow-reactor format was higher than the highest throughputs (e.g., 1500 reactions per day) in the automated synthesis and screening systems reported so far35,36,37, we noticed that it had the potential to be improved further.
Steady-state and non-steady-state experimental mode
During the high-throughput screening experiments for different reaction conditions, when the synthesis and characterization of the previous condition experiment were completed in the flow system, a new reaction solution needed to be introduced into the reactor and detection flow-cell channels. Due to the convection and molecular diffusion effects existed in the flow system, the previous reacted solution and the newly-introduced unreacted solution would mix with each other at their junction region, and the absorbance signals detected by the UV-Vis detector exhibited a dynamically-changing format (Fig. 3A1) during the switching process of different solutions. The conventional method is to wait for the newly-introduced unreacted reaction solution to flush all of the previous reacted solution out of the flow system to enable the detector to obtain a stable plateau-type steady-state absorbance signal for data reading. However, such a steady-state experimental mode requires much longer waiting time for obtaining the steady-state signals. In the above photocatalytic screening experiment under the steady-state mode, the lasers kept irradiating the microreactor channel and the system spent most of the time (27 s of the 32 s of one experimental cycle time) in switching the different experimental conditions and waiting for a steady-state detection signal to be obtained (for example, as shown in Fig. 3A2). Such a waiting time far exceeded the actual time (<4 s) for photocatalytic synthesis and characterization for a reaction solution, severely limiting the screening throughput for different reaction conditions. In fact, this is one of the major limiting bottlenecks in the application of current flow chemistry systems to high-throughput screening.

A Typical recordings of the steady-state and non-steady-state absorbance signals obtained in the screening for S-5, with a photocatalyst ratio of 2 mol% and a S-5 concentration of 0.01 M. (A1) Typical absorbance recordings of 6 different reaction conditions under the steady-state screening mode. (A2) Enlarged view of the absorbance recordings for the first two conditions of the 6 reaction conditions in (A1). It took an average time of 27 s in each condition cycle to obtain the steady-state absorbance signal, which was calculated from the difference between the reacted steady-state plateau absorbance and the unreacted blank absorbance. (A3) Typical absorbance recordings of 30 different reaction conditions under the non-steady-state screening mode. (A4) Enlarged view of the absorbance recordings for the 17th and 18th conditions of the 30 reaction conditions in (A3). Each condition cycle took an average time of ca. 4 s, with a non-steady-state signal peak containing 40 absorbance data points, obtained using the laser pulse irradiation method. B Recordings of the non-steady-state peak signals obtained in the screening experiment of the total 12,000 reaction conditions, which was replicated three times to test the repeatability.
To increase the efficiency of time utilization and screening throughput, we proposed the strategy of non-steady-state experimental mode instead of the steady-state mode by using the laser pulse irradiation method to turn the irradiation laser on and off for achieving the rapid switching between the reacted and unreacted solutions, producing a series of non-steady-state continuous peak-shaped signals as shown in Figure 3A3. Under the non-steady-state mode, the waiting time for reaction solution switching was shortened to 6 s and the average time for each experimental cycle was shortened to 8.5 s, achieving an ultra-high throughput up to 10,000 reaction conditions per day (Fig. 3A3, A4, B).
However, these non-steady-state peak signals included the combined absorbance information of the reactants and products from the previous reacted and the newly-introduced unreacted reaction solutions, which were influenced by multiple factors related to the convection and molecular diffusion effects, such as the reaction solution flow rate, the inner diameters and lengths of the reactor and the detection flow-cell channels, and the reactants and products molecular weights. Therefore, it was a great challenge to acquire the corresponding steady-state absorbance data from the non-steady-state peak signals for evaluating the reaction progress.
AI-assisted absorbance prediction
For achieving this complex and challenging task, we developed the AI-assisted absorbance prediction method by using the AI method to analyze the influencing factors related to the convection and molecular diffusion effects and decoupling the non-steady-state data of the adjacent reaction solutions mixed with each other, to predict the corresponding steady-state absorbance data of the respective reaction solutions.
In order to obtain accurate prediction results, we attempted to use 10 regression models based on the principles of linear models, decision tree, neural networks and integrated learning, to process the large numbers of the non-steady-state absorbance data and to predict the corresponding steady-state absorbance data under the same reaction condition, from which we searched for the best-performing model. In these models, the non-steady-state absorbance data (i.e., 40 absorbance data points recorded for each non-steady-state signal peak, Fig. 3A4) as well as the all corresponding 8 variables (i.e., flow rate, laser light intensity, wavelength, substrate concentration, photocatalyst ratio, photocatalyst concentration, substrate species, and photocatalyst species) of the present flow photocatalytic system were set as the inputs of the models (Fig. 4A). The corresponding steady-state absorbance data obtained experimentally using the steady-state mode with the same conditions (i.e., real steady-state absorbance data) were set as the targets of the models, and the predicted steady-state absorbance data were set as the outputs of the models. The substrate and photocatalyst species were input to the models in the form of relative molecular weights instead of chemical structures, since they are directly related to the molecular diffusion effect. On the basis of the massive amounts of the output and target data of the 12,000 reaction conditions (Fig. 4B1), we evaluated the performance of the 10 regression models based on the R2 and RMSE values of the test set. The RMSE values were calculated based on the targets (i.e., real steady-state absorbance data) and the outputs (i.e., predicted steady-state absorbance data) of the models. Among these models, the XGB regression model demonstrated the best prediction performance (Fig. 4B2), which had the smallest RMSE of 0.0140 and the largest R2 of 0.991 with the 70: 30 proportion of the training and test set. We tried to use less data to predict more data (such as 2.5% of the data as the training set and 97.5% of the data as the test set) to further test the predictive performance of the XGB regression model. Pretty good result was still obtained where 300 conditions data were used to predict the remaining 11,700 conditions with a RMSE of 0.0550 and R2 of 0.859 (Fig. 4B3).
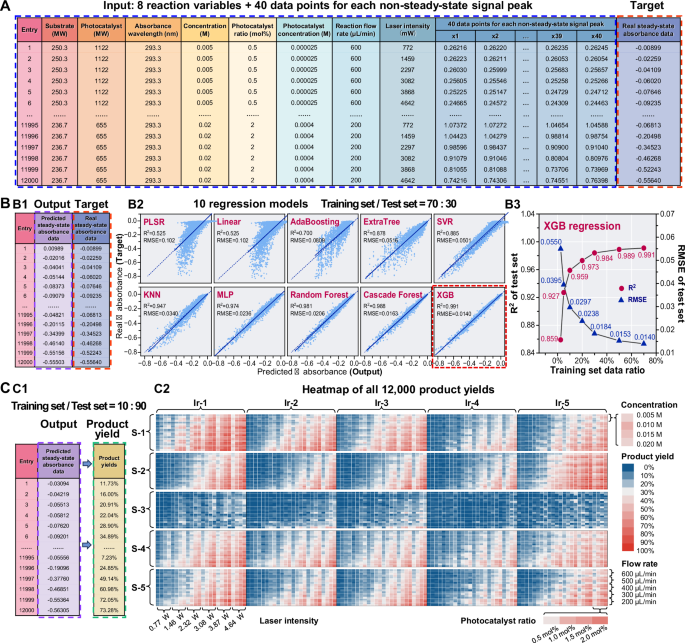
A Composition of the large dataset used in the AI-assisted steady-state absorbance prediction, including the 12,000 absorbance data with 48 eigenvalues (8 reaction variables and 40 data points for each non-steady-state signal peak) and 1 target (steady-state absorbance data). B AI-assisted prediction models for predicting the steady-state absorbance data from the non-steady-state absorbance data. (B1) Predicted and real steady-state absorbance as the outputs and targets of the models, respectively. (B2) 10 different regression models for the prediction of the steady-state absorbance data from the non-steady-state absorbance data, including partial least squares regression (PLSR), linear regression, adaptive boosting (AdaBoosting) regression, extremely randomized trees (ExtraTree) regression, support vector regression (SVR), k-nearest neighbor (KNN) regression, multi-layer perceptron (MLP) regression, random forest regression, cascade forest regression, and extreme gradient boosting (XGB) regression models. The dashed line is the y = x line, and the solid line is a linear fit curve between the predicted and the true steady-state absorbance values. The two metrics, R2 and RMSE values, were calculated using the corresponding functions within sklearn.metrics. (B3) Performance of the test set of the XGB regression model. The prediction accuracy of the test set gradually decreases as the proportion of randomly selected training set data decreases from 70% to 2.5%. C Screening results of the total 12,000 reaction conditions obtained using the AI-assisted steady-state absorbance prediction method. (C1) Predicted steady-state absorbance data output from the XGB model with training set: test set = 10: 90 were converted to product yields. (C2) Heatmap showing the screening results of a total of 12,000 reaction conditions, including the orthogonal combination of 5 substrate species, 5 photocatalyst species, 4 concentrations, 4 photocatalyst ratios, 5 flow rates, and 6 laser light intensities under the non-steady-state mode.
These results showed that with the use of the non-steady-state mode and the AI-assisted absorbance prediction method, the long-standing challenge limiting the improvement of screening throughput of flow chemical screening systems caused by inefficient and time-consuming condition switching could be solved. Correspondingly, the screening throughput for the photocatalytic [2 + 2] cycloaddition reaction conditions increased from 2600 to 10,000 conditions per day using the non-steady-state experimental mode, which is the highest level reported in the field of organic synthesis so far. If the manual mode of conventional batch photocatalytic organic synthesis experiments is used to complete the same workload, it would require 2000 organic synthesizers to work one day or a synthesizer to work 2000 days, assuming that each person could complete 5 condition screening experiments per day. What’s more, we repeated all the non-steady-state experiments 3 times to ensure the repeatability, and all 36,000 peak signals are shown in Fig. 3. In addition, in terms of reagent consumption, only 4.0 mmol of each substrate and 0.05 mmol of each photocatalyst were required to complete the whole screening experiment.
We used 10% of the non-steady-state absorbance data as the training set of the XGB model to predict 12,000 steady-state absorbance data, and a 10-fold cross-validation study was performed and the results are shown in Supplementary Information (Table S9). All the absorbance data were converted into product yields (Fig. 4C1). There are 25 data squares in the heatmap, each of which consists of the data of 1 substrate and 1 photocatalyst condition with different concentrations, photocatalyst ratios, flow rates, and laser light intensities (Fig. 4C2). These data squares show different profiles of the product yield distribution, indicating that the photocatalyst and substrate species have significant impact on the product yields. Among the four variables, although all of them show effects on the product yields, the effects of the flow rate and laser light intensity are more obvious. Therefore, we further present the 12,000 data using multidimensional bubble plots with the x-axis of the flow rate and the y-axis of laser light intensity.
Factors influencing photocatalytic [2 + 2] cycloaddition reaction
In the large majority of experiments, the yield of the reaction products significantly increased with the increase of the laser light intensity (Fig. 5A), indicating that sufficient high light intensity is a necessary prerequisite for the high-efficient photocatalytic [2 + 2] cycloaddition reaction in the LCW photocatalytic microreactor.
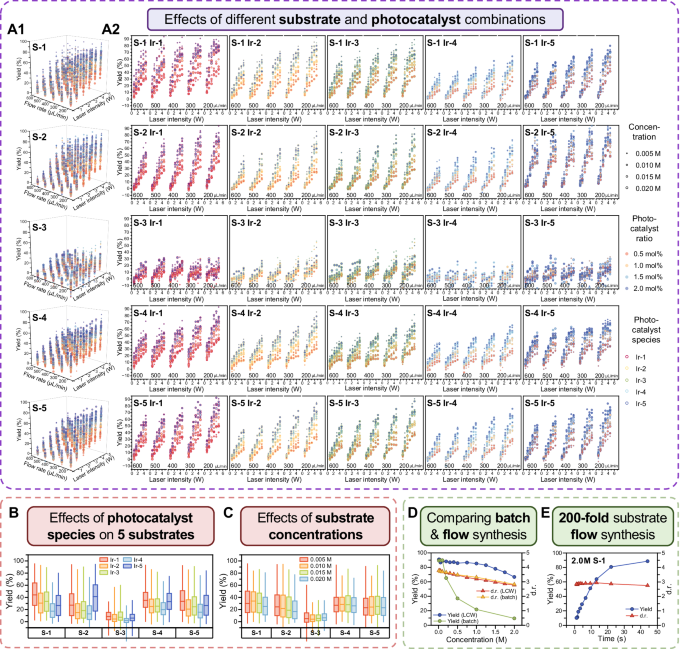
A Multidimensional bubble plots of the 12,000 data, showing the effects of different variables on the product yield, including the species, concentrations, and ratios of the substrates and photocatalysts, laser light intensity, and flow rate of the reaction solutions. Each 6-dimensional bubble plot in (A1) contains 2400 product yield data of 1 substrate specie and 5 photocatalyst species. Each 5-dimensional bubble plots in (A2) contains 480 product yield data of 1 photocatalyst and 1 substrate specie, corresponding to a data square in the heatmap shown in Figure 4C2. In each 5-dimensional bubble plot, the colors of the contour lines of the bubbles represent different photocatalyst species, the colors filled in the bubbles represent different photocatalyst ratios, and the sizes of the bubbles represent different substrate concentrations. B Box plot showing the effect of the photocatalyst species on the product yields of the 5 substrates. The three horizontal lines of each box from top to bottom represent the first, median, and third quartiles of the product yield data, respectively. C Box plot showing the effect of the concentrations of the 5 substrates on the product yields. The three horizontal lines of each box from top to bottom represent the first, median, and third quartiles of the product yield data, respectively. D Comparisons of the variations of the product yield and d.r. with the increase of the S-1 concentration using the batch and present flow methods. E Variations of the product yield and d.r. with the increase of the residence time in the LCW microreactor using S-1 with a high concentration of 2.0 M, which is 200-fold of that in conventional batch systems.
As shown in Fig. 5A, the product yield increased as the flow rate of the reaction mixture decreased. A higher flow rate means a higher throughput while a shorter residence time (i.e., light irradiation time) of the reaction solution in the photocatalytic microreactor, which results in a lower reaction yield (Fig. 5A). The present high-efficiency LCW photocatalytic microreactor could overcome the contradiction between the throughput and product yield, with which the photocatalytic reaction speed could be dramatically increased compared to the batch synthesis methods38,39,40,41, while a high level of product yield and selectivity could also be ensured. For example, in the experiment for S-1, a residence time of 3.3 s in the photocatalytic microreactor (corresponding to the flow rate of 200 μL/min) was sufficient to ensure that the substrate S-1 was completely and selectively converted to the target product.
As shown in Fig. 5B, the optimal photocatalyst for each of the five substrates was quite distinct, which reflects the significance of the high-throughput screening. For S-1, S-4 and S-5, Ir-1 was the optimal photocatalyst with the highest yields, while the photocatalyst Ir-5 was the optimal photocatalyst for S-2 and the photocatalyst Ir-3 was the optimal photocatalyst for S-3.
For the effect of the photocatalyst ratio, generally, the higher the photocatalyst ratio, the faster the reaction speed and the higher product yields (Fig. 5A). For meeting the requirements of green chemistry, 1 mol% photocatalyst is an optimal cost-efficiency choice under a sufficient light intensity.
For the effect of the substrate concentration, in most of the plots (Fig. 5C), we did not observe an evident correlation between the product yields and the substrate concentrations of S-1, S-2, S-3, S-4, and S-5 in the tested range of 0.005 M to 0.02 M, which are frequently adopted in conventional photocatalytic reaction experiments. The reason for this phenomenon may be that our system has an ultra-high catalytic capacity which did not show a significant difference in product yield in the tested range of the substrate concentrations.
In the conventional batch photocatalytic systems, how to increase the substrate concentrations while ensuring sufficiently high yields has been a critical challenge and has not been well addressed so far. For example, in the photocatalytic [2 + 2] cycloaddition reaction reported by Yoon et al. using a conventional batch photocatalytic reactor, when the substrate concentration increased from 0.01 M to 0.05 M, the product yield reduced from 89% to only 33%30. We further increased the concentration of substrate S-1 using 50 μL/min flow rate (i.e., 13.2 s residence time) in the present system, the results showed that the product yield decreased slightly from 91% to 85% when the substrate S-1 concentration increased from 0.01 M to 1.0 M, and further reduced to 67% when the substrate S-1 concentration reached its solubility limit of 2.0 M (Fig. 5D). Compared with that (0.01 M) of the conventional batch system, the substrate concentration could have a 100-fold increase without evidently affecting the reaction yield. We further reduced the flow rate of the 2.0 M substrate reaction solution, the yield could reach 89% at 15 μL/min (i.e., 44 s residence time) (Fig. 5E). The high yields obtained at high substrate concentrations could be attributed to the high light intensity and the fast mass transfer effect in the present photocatalytic microreactor. Such a breakthrough result is of great significance to the application of the photocatalytic cycloaddition reaction in the industrial production of related drugs.
AI-assisted cross-species prediction
To further utilize the above 12,000 data and preliminarily explore the potential possibility of applying AI technique to intelligent chemical synthesis screening, we used the XGB algorithm to perform AI-assisted prediction of product yields cross-substrate and cross-photocatalyst. For cross-species prediction of product yields, the inputs of the models were the reaction conditions (i.e., the 8 reaction variables), the targets were the product yields obtained experimentally, and the outputs were the predicted product yields (Fig. 6A). Differing from the AI-assisted absorbance prediction, the cross-species prediction required detailed chemical structure information of the substrate and photocatalyst species, which were described, digitized, and input to the models using the SMILES strings to generate Modred descriptors (Table S1). The Mordred descriptor dimensions of each substrate and photocatalyst species were reduced to 2 in order to match the dimensions of the other variables and facilitate visualization in subsequent study, as described in Methods. The inputs consisted of 10 dimensions, with 2 representing the substrate species, 2 representing the photocatalyst species, and the remaining 6 representing the other variables (Fig. 6A).
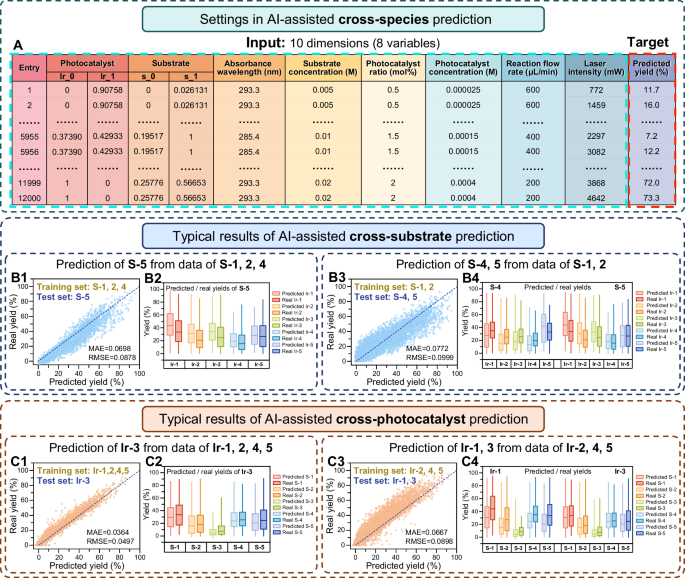
A Settings used for AI-assisted cross-species prediction, including inputs and targets of the models. B Typical results of the AI-assisted cross-substrate prediction, including prediction of S-5 from data of S-1, S-2, and S-4 (B1), and prediction of S-4, S-5 from data of S-1 and S-2 (B3) with box plots showing product yields results (B2, B4), respectively. C Typical results of the AI-assisted cross-photocatalyst prediction, including prediction of Ir-3 from data of Ir-1, Ir-2, Ir-4 and Ir-5 (C1), and prediction of Ir-1, Ir-3 from data of Ir-2, Ir-4 and Ir-5 (C3) with box plots showing product yields results (C2, C4), respectively. The model performance metrics for the cross-validation studies include the MAE and RMSE values. The entire results of the cross-species prediction are shown in Fig. S11, Table S10, S11.
Both the results of the cross-species prediction with different training set ratios and the cross-validation studies are described in the Supplementary Information (Fig. S11, Table S10, S11). The 12,000 experimental data were divided into training and test sets with different ratios for AI-assisted cross-species prediction. As a typical result of the cross-substrate prediction, we used the data of S-1, S-2, and S-4 as the training set to predict the yields of S-5, achieving MAE = 0.0698 and RMSE = 0.0878 (Fig. 6B1). The distinct effects of the 5 photocatalyst species on S-5 were accurately predicted and Ir-1 was predicted as the optimal photocatalyst, which is consistent with the experimental results (Fig. 6B2). With a smaller training set of S-1 and S-2, the prediction for S-4 and S-5 achieved with MAE = 0.0772 and RMSE = 0.0999 (Fig. 6B3, B4). For the cross-photocatalyst prediction, the data of Ir-1, Ir-2, Ir-4 and Ir-5 could be used to predict the yields of Ir-3, with MAE = 0.0364 and RMSE = 0.0497, which presented similar results to the real product yields (Fig. 6C1, C2). When the training set was reduced to include three photocatalysts of Ir-2, Ir-4 and Ir-5, pretty good prediction for Ir-1 and Ir-3 could still be obtained with MAE = 0.0667 and RMSE = 0.0898 (Fig. 6C3, C4). These surprising results exemplarily demonstrated the attractive possibility of utilizing AI algorithms for cross-substrate and cross-photocatalyst prediction of organic synthesis reactions.
link





.jpg)