Photo-CIDNP for quantification of micromolar analytes in urine
Experiment optimization and matrix effect on photo-CIDNP
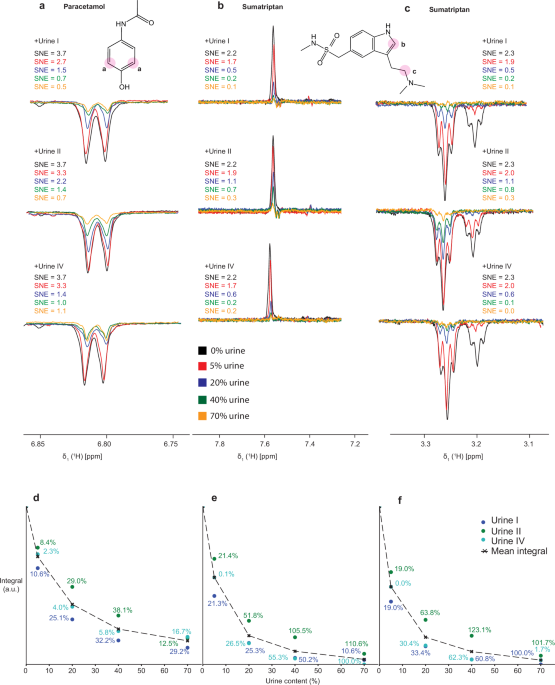
In our experiments, we considered the resonances at 6.8 ppm for paracetamol and at 7.6 and 3.2 ppm for sumatriptan. The sign of the peaks calculated by the Kaptein rules suggested that the hyperpolarization of paracetamol originates from the recombination product, while for sumatriptan it originates from the escape product (Supplementary Equation 1, Supplementary Table 1). Since the singlet excited state of fluorescein is short-lived, the photo-CIDNP signal likely comes from a triplet precursor32.
As a proof of concept in a realistic situation with variable biofluid composition, we performed experiments on a set of six different human urine samples obtained from healthy volunteers. We started with optimizing the sample conditions, including the dye concentration, pH of the solution, and number of acquired scans. We assessed four different dyes, fluorescein, 4’,5’-dibromofluoresein, riboflavin and flavin mononucleotide (FMN) for best suitability. Among these, fluorescein was most resistant to photo-bleaching, gave by far the highest enhancement and exhibited the highest absorbance at our wavelength of 450 nm (Supplementary Figs. 1,2), hence it was chosen for all further studies. The photo-CIDNP activity of the target compounds was tested at fluorescein concentrations of 25–100 μM, not showing any significant variation (Supplementary Fig. 3). Then, we tested the influence of pH in the range 5–8 (Supplementary Fig. 4). For sumatriptan and paracetamol, the photo-CIDNP enhancement dropped by 20–30% at low pH (pH 5–6) compared to physiological pH (pH 7.1–8). For the high field measurements at 14.1 T field strength, we found that 64 scans delivered the optimal total sensitivity, as the photo-CIDNP enhancement is non-linear due to radical reactions and photobleaching (Supplementary Fig. 5). For the low field measurements at 1.9 T field strength, optimal results were achieved with 32 scans. It is important to note that for samples containing 100 μM of paracetamol, conventional spectra measured at 1.9 T did not provide detectable signals at 32 scans. The signal-to-noise ratio (SN) obtained in 32 scans laser experiment is comparable with dark spectrum measured with 10,000 scans (Supplementary Fig. 6). To perform the analysis in biofluids, we used lyophilized urine samples to prevent changes over time and reconstituted them immediately before measurements in either H2O or D2O. We verified that the lyophilization did not affect the photo-CIDNP performance by comparing the photo-CIDNP-enhanced signal for a reference compound in freshly collected urine and lyophilized urine (Supplementary Fig. 7). As a reference signal for quantification, we used 5-fluoro-2-methylindole, which features a particularly high photo-CIDNP effect. This reference compound provides the additional benefit that its most enhanced signal at 6.15 ppm is well separated from the complex signal array of the urine background matrix.
To investigate the matrix interference effect on the photo-CIDNP performance, for both analytes, paracetamol and sumatriptan, we measured samples with increasing urine content from 0 to 70% (Fig. 1). The photo-CIDNP effect was analyzed as the signal-to-noise enhancement22 (SNE, Supplementary Equation 2) for the peak at 6.8 ppm for paracetamol and for the peaks at 7.6 and 3.2 ppm for sumatriptan. Compared to the buffer, we observed a drop in photo-CIDNP enhancement by 80–95% upon adding 70% of urine to the sample. Even only 5% urine caused a severe photo-CIDNP signal decrease of around 30% for all tested urines and both analytes. Importantly, the drop of the hyperpolarized signal was not uniform across different urines and for the different analytes, indicating that the particular urine composition had a specific impact on the signal enhancement. This finding thus implies a specific and strong matrix interference on the photo-CIDNP performance. However, at 5% urine content, the variation in photo-CIDNP enhancement drop between different urine samples was much less pronounced than at higher urine concentrations, reaching maximally a deviation from the mean of 20% (Fig. 1d–f). In contrast, the deviation reached over 100% at high urine content. This suggests sample dilution could be a way to overcome the matrix interference effect partially and to apply a universal calibration curve for highly diluted samples.
a 1D 1H photo-CIDNP NMR spectra of paracetamol in different dilutions of urine. The corresponding SNEs is indicated, starting from 0% (sample measured in pure buffer) to 70% of urine. The position of the resonance is indicated by pink circles on the structure. b, c Same for two sumatriptan signals. Spectra were recorded with 64-scan gated illumination photo-CIDNP at 14.1 T with a 0.2 s laser irradiation block. Samples contained 100 μM of the analyte and 100 μM of fluorescein in 100 mM sodium phosphate buffer at pH 7.1. SNEs were calculated relative to the dark spectrum measured with modified W5 and no urine addition. d–f Dependance between the urine content and the photo-CIDNP active signal integral with the percentage deviation from the mean at each urine content for each resonance, respectively.
To study the nature of the matrix interference effect further, we probed the effect of the oxygen removal system. Two samples were prepared using either the enzymatic scavenging system or conventional nitrogen degassing. We found no difference in signal enhancement between the two systems for samples containing 40% urine and 5-fluoro-2-methylindole (Supplementary Fig. 7), leading to the conclusion that the observed signal enhancement drop is not dependent on the oxygen scavenging system. Human urine contains a small fraction of large proteins53 to which small molecules could potentially bind in a specific or non-specific fashion, becoming unavailable for photo-CIDNP. We removed proteins with molecular weight larger than 3 kDa by ultra-filtration, but we observed no difference in signal intensities between filtrated and non-filtrated urine (Supplementary Fig. 7), thus ruling out dye- or compound-binding as a potential cause of the observed photo-CIDNP drop. Therefore, we concluded that the source of the photo-CIDNP drop is a competition effect between the photo-CIDNP active molecules and potential triplet state quenchers that might be a part of the biological matrix. Moreover, the photo-CIDNP active target molecule is most likely also affected by exchanging electrons in its neutral or radical state with other molecules by reductive electron transfer. Trying to resolve which are the major contributors to the photo-CIDNP drop, we investigated a series of samples containing 40% of a solution of individual urine components, including creatinine, urea, thiamine, d-mannitol, succinate, formate, pantothenic acid, glucuronate, cytosine, sodium pyruvate, nicotinamide, bilirubin, a mixture of amino acids, uric acid, lactic acid and salts mixture, in the urinary excretion range54 (Supplementary Table 2, Supplementary Fig. 8). In photo-CIDNP enhanced spectra we observed a 68% signal drop upon uric acid addition, 30% upon salts, 20% upon bilirubin and 16% upon amino acids. Notably, uric acid was buffered to pH 7.4 in 100 mM sodium phosphate buffer, and this pH value should not have affected the photo-CIDNP enhancement. The mix of all individually tested components led to a photo-CIDNP enhancement drop of 30%, which does not fully match the value observed in 40% urine. The non-additivity of the single influences implicates the presence of complicated interactions between the mixture components and confirms that the problem arises from multiple species, affecting the pool of fluorescein and analyte.
We specifically tested bilirubin due to its structural similarity to urobilin, a compound typically present in urine54. The absorption spectrum was recorded for bilirubin (Supplementary Fig. 2). As bilirubin absorbs at the excitation wavelength used in our experiments, it may act as an efficient triplet quencher. To assess whether the removal of colored compounds such as urobilin could enhance the photo-CIDNP signal, by reducing triplet quenching and minimizing competition for light absorption, we photobleached urine samples using a 600 mW laser at 450 nm for 256 s (Supplementary Fig. 9). Absorption spectra were then compared before and after photobleaching, along with the photo-CIDNP spectra of samples containing 40% urine and the reference compound 5-fluoro-2-methylindole. The results showed only a negligible improvement in photo-CIDNP enhancement following photobleaching. This indicates that although eliminating colored substances can slightly enhance the signal, they are not the primary contributors to signal loss in complex matrices. These findings support our earlier conclusion that part of the photo-CIDNP signal loss in complex matrix is due to the presence of bilirubin.
To ensure that the differences in photo-CIDNP enhancement do not stem from altered fluorescein absorbance, we spiked three samples, 40% urine I, II, or IV, with 100 μM fluorescein and measured their absorption spectra (Supplementary Fig. 10). These were then compared to a reference sample containing fluorescein in pure buffer. We observed negligible differences in absorbance at 450 nm, indicating that the urine matrix at 40% concentration does not affect the absorption properties of fluorescein.
Evaluation of NMR experiments for photo-CIDNP in urine
To develop methods for analyte quantification, we produced mock patient samples where urine was enriched with a known amount of sumatriptan or paracetamol in the concentration range of 12.5–250 μM, matching the natural urinary excretion range55,56. Samples were then diluted with buffer to 40% urine. At this urine content, we could still observe photo-CIDNP signal enhancement. We measured non-irradiated (dark) modified perfect-echo W5 experiments and gated illumination experiments (laser) for samples containing 40% urine without adding external photo-CIDNP active compounds to investigate the variability in the used urine compositions (Supplementary Fig. 11). As expected, we observed considerable spectral differences between the urine samples, both in the dark spectra and in the photo-CIDNP active molecules. Various photo-CIDNP active compounds natively occurring in urine further support the hypothesis that the competition effect affects the photo-CIDNP performance of the target molecule.
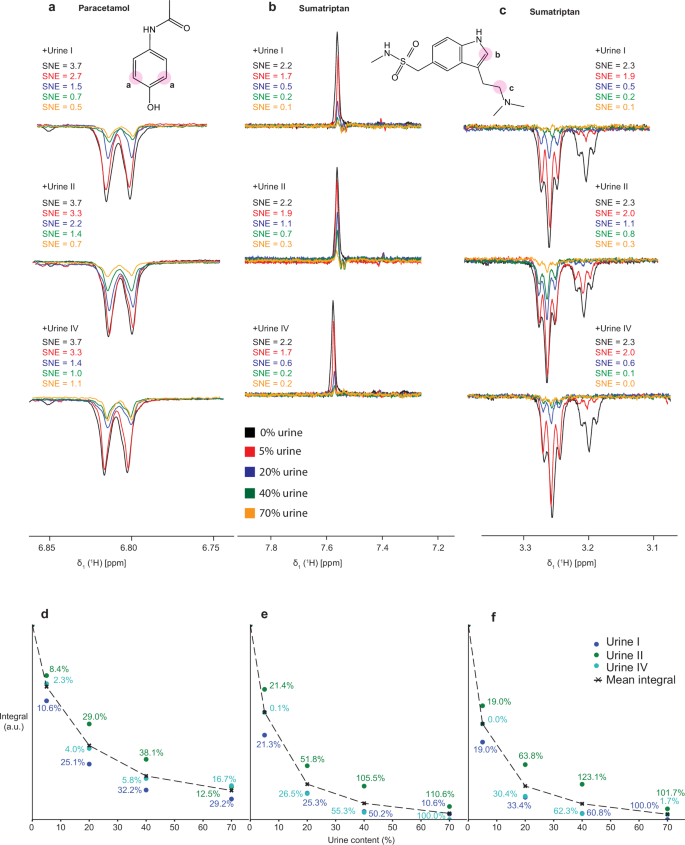
To evaluate the two NMR experiments used for detection, modified perfect-echo W5 water suppression and gated illumination, we collected spectra from samples with 100 μM paracetamol and/or 100 μM sumatriptan (Fig. 2, Supplementary Fig. 12, 13). We measured the spectra of a single analyte in buffer, and with the addition of 40% of urine I, II, or IV. We also verified that none of the urine samples contained the analytes. First, by comparing spectra of pure analytes in buffer, we noted that, although gated illumination helps to extract signals corresponding to photo-CIDNP active species and reduces spectral complexity, it results in around 2.5 times lower SNE than the standard experiment based on the modified W5 water suppression. The background removal by gated illumination might not seem crucial for samples containing 100 μM paracetamol, however, at lower analyte concentrations, the signal of interest would overlap with background signals in modified W5 water suppression experiments and can only be detected by gated illumination (Fig. 2). Furthermore, for this type of experiment, similarly like in work presented by Kuhn et al.45, we found that the reconstitution of urine in D2O provided better background nulling and was thus further used as the solvent.
a 1D 1H NMR spectra of 100 μM paracetamol in pure buffer measured in dark mode (no photo-CIDNP), laser mode (1 s of laser illumination) and with gated illumination (0.2 s of laser irradiation). b–d Same after doping samples with 40% of urine I, II, or IV, respectively. Black spectra are reference spectra of the urine background without paracetamol. All spectra were measured at 14.1 T with 64 scans. SNEs were calculated in comparison to the dark spectrum measured with modified W5 and no urine addition.
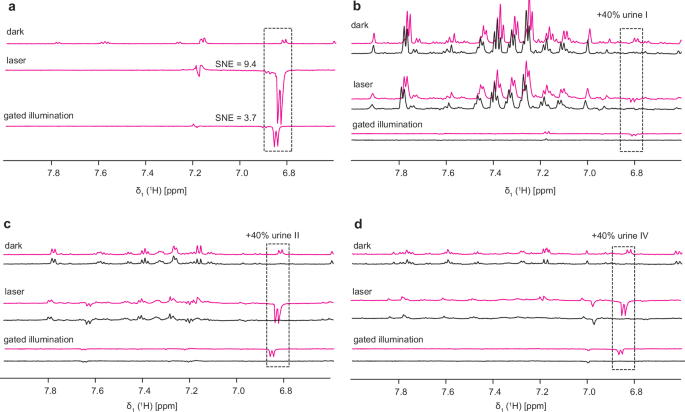
As a step towards quantification of analyte concentration, we next tested the use of a reference compound that would serve as a concentration standard (Fig. 3). We examined samples containing 40% of four different urines and the analytes of interest. As possible concentration standards, we tested two different compounds, 5-fluoro-2-methylindol and 2-(2-amino-1,3-thiazol-4-yl)propan-2-ol as internal references. Both were chosen due to their high photo-CIDNP activity, the former verified by us, the latter known in the literature22. We tested 1:1 mixtures (100 μM) of analytes with internal references, including a 1:1 mixture of paracetamol and sumatriptan. The study showed that the ratio of the integrated signals for the analyte and reference varied significantly between different urine matrices. This suggests that native urine components influence the photo-CIDNP enhancement of the target and reference molecules in distinct ways, likely by competing for the dye or by electron transfer between radicals. In some cases, the photo-CIDNP-enhanced signals for sumatriptan could not be detected anymore in the spectrum. This highlights the sample composition’s influence on the gained polarization. Unfortunately, these findings excluded the option of an internal reference application due to the massive non-linearity of the matrix interference effect. To circumvent this problem, we developed two methods that bring back linearity and thus allow the quantification of analytes by photo-CIDNP in complex mixtures. We discuss these two methods in the following paragraphs.
a Chemical structures of analytes (paracetamol, sumatriptan) and internal references (5-fluoro-2-methylindole and 2-(2-amino-1,3-thiazol-4-yl)propan-2-ol) with highlighted protons used in analysis. b–d Photo-CIDNP 1D 1H NMR gated illumination spectra of samples containing analytes. Pink spectra contain 100 μM of one analyte, black spectra contain analyte, reference compound, and 40% of urine I–IV. The integral ratios of the analyte (pink dashed box) over the reference (black dashed box) signal are given. Spectra were recorded with 64 scans and 0.2 s of laser irradiation at 14.1 T.
Method 1: quantification with spiking
For the spiking approach, a given sample with analyte concentration in the 12.5–250 μM range was split into two halves. One half of the sample remained unmodified, and the other half was spiked with a known amount of the analyte. From the signal difference of the two samples, the initial concentration can be calculated under the assumption that the signal depends linearly on the concentration in the concentration range spanned by the spiking. We tested this approach for paracetamol and sumatriptan as target analytes in samples containing 40% urine using gated illumination experiments, measuring each point in triplicates (Fig. 4 and Supplementary Fig. 14). The data show that the relation between the photo-CIDNP signals and the analyte concentration is indeed linear in the examined concentration range (Fig. 4a, Supplementary Fig. 14a). Notably, the slope had a strong variation between different urine samples, corresponding to vastly different compositions of the biofluids. For the quantification, we chose peaks with high SNE enhancements. For paracetamol, the signal at 6.8 ppm corresponding to aromatic protons in the ortho position, and for sumatriptan, the methylene signal at 3.2 ppm was selected (Fig. 4b, Supplementary Fig. 14b). Spiking was performed for four native concentrations in the range of 12.5–250 μM in three different urines, at 25, 75, 125, and 150 μM.
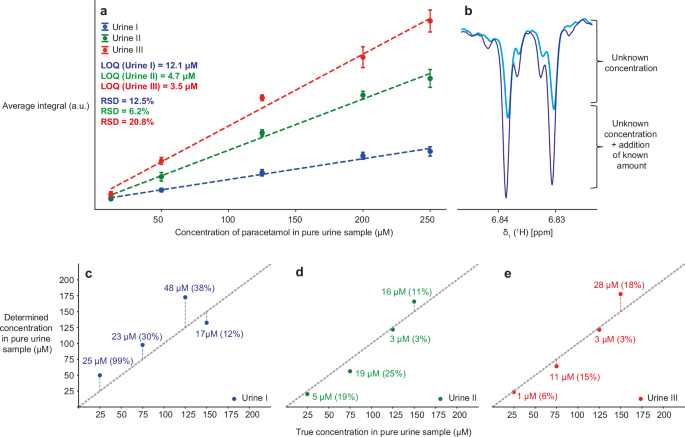
a Calibration curves for three sets of 40% urine I, II, III with paracetamol concentration of 12.5–250 μM (before dilution, so 5–100 μM after dilution). Each point corresponds to the average integral from triplicated experiments. The indicated LOQ corresponds to SN = 3 in native urine samples. RSDs are calculated for the points with the lowest analyte concentration (12.5 μM before dilution). b NMR signal intensities for paracetamol for an unknown concentration (light blue) and after spiking with 20 μL of 0.5 mM stock solution (dark blue). c–e Correlation of the experimentally determined with the true concentration in three different urines I–III. The difference between the true and the determined concentration is indicated next to each point with the percentage error in the brackets. All photo-CIDNP 1D 1H NMR experiments were measured at 14.1 T, using gated illumination with an irradiation time of 200 ms and 64 scans.
LOQ were estimated based on the SN of individual peaks. Instead of using the conventional SN threshold of 10, we applied a lower threshold of 357,58,59,60. This choice was justified by the high measurement precision observed, as reflected in the relative standard deviation (RSD) values58. Due to the limited volume of biological samples available, RSDs were calculated at the lowest concentration point in the calibration curves rather than directly at the LOQ. In most cases, the RSDs were below 20%, which falls within the acceptable range for analytical tests in the early stages of development58. In a few cases where the RSD exceeded 20%, we still used an SN of 3 for LOQ estimation. This was due to the current experimental setup introducing greater signal variability, as discussed in the following section. Importantly, these deviations are not fundamental limitations and can be minimized through improvements in the setup as explained in the next paragraph.
Because different urines affect photo-CIDNP to varying degrees, the LOQ depend significantly on the sample used as well as the photo-CIDNP activity of the measured analyte, with LOQs ranging from 3.5 to 12 μM for paracetamol and 13 to 42 μM for sumatriptan in native urine samples (before dilution). By measuring the experiments in triplicate, we quantified the experimental variation of the measurements, which resulted from our technical setup at 14.1 T. In this setup, illumination is applied from the top, and the inevitable slight variations in the fiberglass position then lead to a difference in illumination at the individual samples. Because the spiking method depends on the comparison of two samples rather than an internal standard, errors induced by this mechanical variation accumulate. Nonetheless, the accuracy of the spiking method was below 30% for most samples and reached errors of below 5% for some samples (Fig. 4c–e, Supplementary Fig. 14c–e). It can thus be expected that a mechanically improved setup, such as a fixed illumination source, will readily eliminate this error source and lower the RSDs. This could be achieved, e.g., by introducing laser optics from the bottom of the spectrometer, in-situ illumination by specialized NMR devices, or including LED light sources inside the probe head37,61.
Method 2: quantification with dilution method
An alternative quantification method was developed based on the observation that the variation in the photo-CIDNP enhancement between different urine samples was much smaller when the urine content was diluted to 5% compared to samples with higher urine concentrations. At 5% urine, the photo-CIDNP signal enhancement is reduced compared to pure buffer, but the variation between different urines is much less pronounced. The non-linear matrix effects, which result from bimolecular reactions, are significantly diminished compared to the overall effect, which scales with the dilution (Fig. 1d–f). The slopes and intercepts of the calibration curves for spiking at 5% urine are thus nearly identical for different urines, in contrast to those obtained at 40% urine (Fig. 5a, b). We worked on samples containing 5% of urines IV-VI, with original analyte concentrations of 20–200 μM (Fig. 5). The high dilution enabled the enhanced perfect-echo W5 water suppression experiments, offering greater sensitivity than gated illumination. Each point of the calibration curve was measured in triplicate. For paracetamol, we selected the same enhanced peak (6.8 ppm) as for the spiking method at high magnetic field (Fig. 5c). For sumatriptan, a peak at 7.6 ppm was used for quantification because of signal overlap with peaks originating from thermal equilibrium (Fig. 5d). By calculating the SN for all points in the calibration curves and then determining the concentration at SN = 3, we determined LOQ, as the RSDs are usually below 20%57,58,59,60. The LOQ for the undiluted urines was in range 6.4–7.1 μM for paracetamol and 15–19 μM for sumatriptan. We then determined the average slopes and intercepts towards a universal calibration curve for each analyte at each magnetic field. Analysis of the errors showed that almost all samples had percentage errors lower than 30%, and half of the samples had a percentage error lower than 10% (Fig. 5e, f).
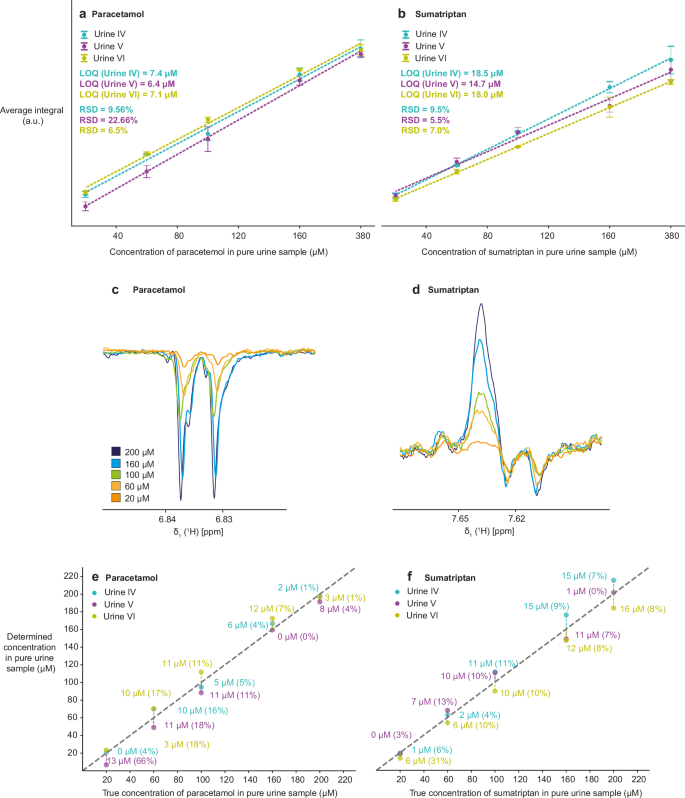
a,b Calibration curves for paracetamol and sumatriptan in range 20–200 μM (before dilution, so 1–10 μM after dilution) in three different urines. The indicated LOQs correspond to SN = 3 in native urine samples. RSDs are calculated for the points with the lowest analyte concentration (20 μM before dilution). All points were measured in triplicates using W5 water suppression, 1 s of laser irradiation, and 64 scans at 14.1 T. c,d 1D 1H NMR spectra at five different analyte concentrations. e,f Correlation of the experimentally determined with the true concentration in 5% three different urines IV–VI. The difference between the true and the determined concentration is indicated next to each point with the percentage error in the brackets.
For future use in low-cost settings, we also verified this approach on a benchtop spectrometer with a low magnetic field of 1.9 T. At this field, paracetamol showed a strong signal enhancement by photo-CIDNP, whereas sumatriptan was not strongly photo-CIDNP active (Supplementary Fig. 15). We worked with concentrations of paracetamol in native urine in the 60–200 μM range and diluted to 5% (Fig. 6a). The peak at 7.3 ppm was used to quantify the signals, and intensities were used instead of integrals, as this resulted in smaller overall errors (Fig. 6b). The determined LOQ ranged from 40–47 μM for the undiluted urine samples. The percentage errors in the low magnetic field calculated for universal calibration curves were comparable to those in the high magnetic field. However, the calculated RSDs are significantly lower than 20%, confirming that the improved setup with a fixed laser position enhances the precision of the measurements compared to those performed in a high magnetic field, where laser illumination via a fiberglass cable from the top results in slight variations in illumination efficiency between measurements. The benchtop setup demonstrates that this issue can be easily avoided.
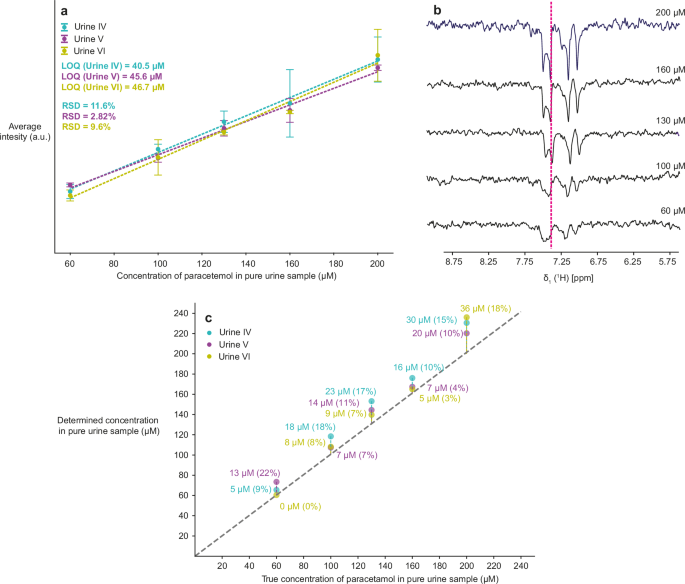
a Calibration curves for paracetamol in range 60–200 μM (before dilution, so 3–10 μM after dilution), in three different urines. The indicated LOQs correspond to SN = 3 in native urine samples. RSDs are calculated for the points with the lowest analyte concentration (60 μM before dilution). All points were measured in triplicates using W5 water suppression, 1 s of laser irradiation, and 32 scans at 1.9 T. b 1D 1H NMR spectra at five different paracetamol concentrations for samples diluted to 5% urine V with the signal used for analysis indicated by the dashed line. c Correlation of the experimentally determined with the true concentration in three different urines I–III. The difference between the true and the determined concentration is indicated next to each point with the percentage error in the brackets.
Comparing the two methods established for analyte quantification, the dilution method was more convenient than the spiking, as it requires the measurement of only one sample instead of two. At the same time, it provides smaller errors, reaching an average below 11% for dilution compared to 26% for spiking. Since the limit of quantification represents a compromise between the matrix effect, which reduces photo-CIDNP enhancement, and the amount of analyte introduced with the urine sample, the appropriate quantification method (spiking or dilution) should be chosen based on the strength of matrix interference and photo-CIDNP activity. For samples that do not exhibit strong quenching effects, spiking appears to allow for lower limits of detection compared to dilution methods (e.g., spiking for paracetamol in Urine II and III versus dilution in Urine IV–VI). For sumatriptan, both methods appear to yield comparable results. The best suitable quantification method should therefore in each case be chosen dependent on the analyte concentration, photo-CIDNP activity, and strength of the matrix interference effect.
Conclusions and outlook
This paper reports the application of photo-CIDNP NMR for the quantitative detection of analytes in complex biofluids, with potential future extension to targeted diagnostics. For the first time, we demonstrate that photo-CIDNP-enhanced signals can be successfully quantified in complex biofluids using two strategies: spiking and sample dilution. The presented quantification methods are effective at both low and high magnetic fields, achieving LOQs as low as 3.5 μM for common drug analytes in real biofluid samples. These results serve as a proof of concept and also as a basis for further development and signal improvement.
As we showed, the matrix interference effect presents, however, a significant limitation in achieving satisfactory photo-CIDNP enhancement in complex biofluids. Complex mixtures lead to a noticeable reduction in photo-CIDNP enhancement compared to pure buffer. For instance, for some samples containing 40% urine, the signal intensity is only slightly higher than that measured without laser irradiation. The intensity is thus also highly dependent on the urine composition. Overall, the photo-CIDNP performance in biofluids still needs to be optimized to get an effective sensitivity enhancement compared to conventional NMR.
In contrast, samples containing only 5% urine show a more significant enhancement, as the concentrations of the quenching components of the matrix decrease with dilution. In parallel, high dilution also reduces the contribution of the thermal signals, such that the gated illumination experiments can be replaced by standard NMR experiments with irradiation block. Standard NMR measurements are feasible in 100% urine samples, but the signal overlap caused by the matrix typically hinders accurate quantification.
A primary advantage of photo-CIDNP thus remains the reduction of spectral complexity. In conventionally measured spectra without laser irradiation, certain signals are undetectable, as they overlap with other signals. In biofluids, gated illumination experiments reduce the spectral complexity, which shown by Kuhn et al.45 and in the present study. This could be further enhanced by exploiting the high specificity of the dye–target interaction. By using specialized dyes with tuned irradiation wavelengths or by adjusting the solution’s pH to influence the ionization of the analytes62, one can steer which signals in the samples will be enhanced. Photo-CIDNP could thus be tuned into a method to obtain completely different spectra from the same biosample just by varying experimental conditions.
A further major advantage of photo-CIDNP is its utility for quantification at low magnetic fields, resulting in significant time savings, mostly because photo-CIDNP hyperpolarization becomes more pronounced as the magnetic field strength decreases22,40. This clearly indicates that while photo-CIDNP has great potential for routine diagnostic applications, further optimization is required. Identifying new dyes and target molecules that are more resistant to matrix interference is crucial, as the matrix effect does not affect all molecules equally. This has been shown in our systematic studies involving mixtures of two photo-CIDNP-active components in complex matrices.
Another important matter is the space of potential target analytes in the future diagnostics tests. One limitation of photo-CIDNP is that not all molecules are active. Active molecules typically contain aromatic structures, as these facilitate stabilizing interactions with the dye through π–π stacking and allow for effective radical stabilization. Therefore, extensive screening of photo-CIDNP-active compounds and in-depth studies of the underlying enhancement mechanisms are essential for identifying potential diagnostic targets. To support this effort, screening studies such as the one presented by Torres et al.29 or Tada et al.63, and the development of technologies that enable high-throughput photo-CIDNP measurements22,29 or connection to microfluidics systems64 are of great importance. Fortunately, many analytes and pharmaceutical compounds contain aromatic systems, making them promising candidates for photo-CIDNP-based detection. According to the studies65,66, significant proportion (80%) of the approved drugs contain aromatic rings, making them potential targets to photo-CIDNP-supported analysis. At the same time, only about 20% of metabolites contain aromatic rings, however, among these are important compounds for diagnostics, such as amino acids, microbial metabolites, and aging biomarkers67,68. As presented by Kuhn et al.45, photo-CIDNP can also be observed in serum, which may facilitate analyte quantification by universal calibration due to the more consistent composition of serum across individuals, unlike urine69.
Future photo-CIDNP-based tests will compete with state-of-the-art diagnostic methods such as LC-MS/MS, GC-MS, HPLC coupled with UV or fluorescence detection, immunoassays, enzymatic assays, and spectrophotometry70,71. The main limitation of photo-CIDNP-based diagnostics, compared to those methods, is sensitivity. While photo-CIDNP can achieve detection limits in the nanomolar range22,36 in the absence of matrix effects, it generally falls short of the sensitivity offered by other currently used techniques, which enable detection at pico- or femtomolar concentrations70. However, the key advantages of photo-CIDNP include very rapid data acquisition (from seconds to few minutes), providing high-throughput measurements, and the much lower cost of benchtop spectrometers and laser setups compared to instruments like mass spectrometers or HPLC systems22. Additionally, benchtop spectrometers are user-friendly, do not require highly qualified personnel to operate, and require barely any maintenance22. In combination with a flow system64, fast quantification of analytes that demand extensive control will become possible, e.g., in the kinetic part of clinical trials or for drugs with narrow therapeutic windows.
To conclude, photo-CIDNP offers substantial advantages, especially when measured at low magnetic fields. With appropriate strategies to mitigate matrix effects—whether through the use of more robust dyes or optimized sample preparation—the technique could become significantly more effective45. Nonetheless, a more systematic investigation of matrix effects is essential for further development of the diagnostic strategies based on the photo-CIDNP hyperpolarization. The approaches developed here have the potential to advance targeted diagnostics by combining the high stability of NMR measurements with the low cost of benchtop instruments and their versatility for various targets and samples.
link





.jpg)