Experimental investigation of anionic and cationic surfactants performance in clay-rich sandstones

CMC measurement
The critical micelle concentration (CMC) can be identified in electrical conductivity diagrams by an evident change in the curve slopes. Below the CMC, increasing surfactant concentration results in significant physicochemical changes, while above the CMC, surfactant molecules form micelles rather than spreading further, stabilizing the properties being measured. The CMC is a vital parameter that indicates when surfactants begin to aggregate and can notably affect their performance in various applications. For cationic surfactant CTAB, the CMC was measured at 1 mM, indicated by the turning point in Fig. 6, where the slope shifts, showing the onset of micelle formation.
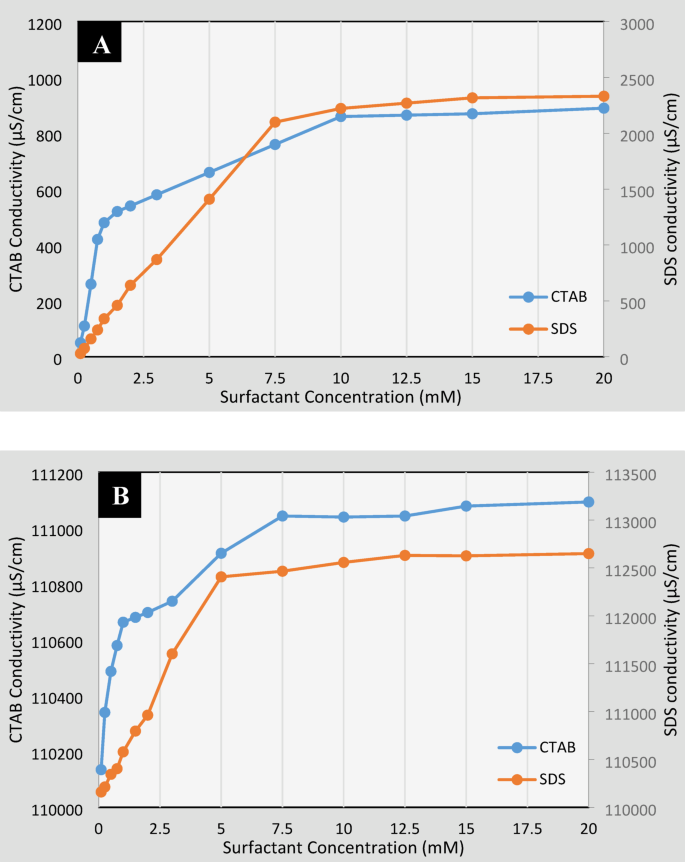
CMC determination plot for CTAB and SDS surfactants in (A) distilled water and (B) in formation brine.
After this concentration, CTAB molecules tend to form saturated structures, leading to a marked reduction in electrical conductivity changes. In contrast, the CMC for anionic surfactant SDS is around 8 mM, indicating that SDS requires a higher concentration to form micelles due to differences in structure and ionic charge. In brine, the CMC values for CTAB decreased slightly to 0.95 mM (5% reduction) and for SDS to 6 mM (25% reduction), highlighting how salinity enhances surfactant aggregation efficiency. These findings underscore the importance of selecting appropriate surfactants based on their CMC values, as they directly influence performance during industrial processes.
IFT results
IFT between oil and water plays a crucial role in determining the efficiency of EOR processes, as it directly impacts the mobility of oil within the reservoir. The results of IFT measurements for CTAB and SDS surfactants are presented in Fig. 7. With increasing CTAB concentration, a notable reduction in IFT was observed. At 0.5 mM CTAB, the IFT dropped to 8.1 mN/m, indicating an initial but partial reduction in surface tension. As the concentration reached 1 mM, close to the CMC of CTAB, IFT sharply decreased to 1.1 mN/m, demonstrating the effect of micelle formation in significantly lowering surface tension. Beyond this point, at concentrations of 3, 6, and 10 mM, IFT values stabilized at approximately 0.9 mN/m. This stabilization suggests that the interface had become saturated with surfactant molecules, achieving optimal IFT reduction.
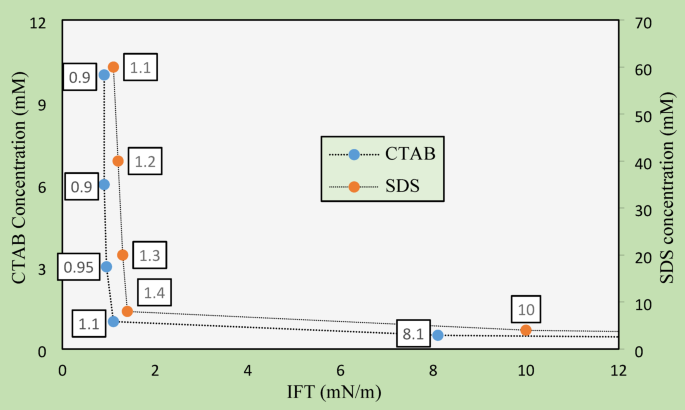
Oil/water IFT results obtained from different aqueous solutions.
In comparison, SDS displayed similar behavior but required higher concentrations to achieve comparable results due to its anionic nature. At 4 mM SDS, IFT was measured at 10 mN/m, showing only a moderate reduction in surface tension. As the SDS concentration increased to 8 mM (approaching its CMC), the IFT decreased to 1.4 mN/m. This reduction highlights the effectiveness of micelle formation in reducing IFT, albeit slightly less efficiently than CTAB. Distilled water and brine solutions, in the absence of surfactants, exhibited significantly higher IFT values of 43 and 45 mN/m, respectively, underscoring the necessity of surfactants in achieving meaningful reductions in IFT for industrial applications, such as oil recovery or emulsion preparation.
These results emphasize the critical impact of surfactant selection and concentration on reducing IFT and enhancing oil recovery. CTAB demonstrated superior performance, achieving lower IFT at a lower concentration due to its cationic nature and stronger interaction with water–oil interfaces. While SDS was also effective, its higher required concentrations and slightly higher IFT levels indicate a less efficient performance. Overall, the findings confirm that optimizing the concentration and type of surfactant is essential for maximizing oil recovery, enabling the effective reduction of IFT under reservoir conditions.
The IFT results demonstrated that CTAB was highly effective in reducing the oil–water IFT, reaching 1.1 mN/m at its CMC (1 mM), which aligns with typical cationic surfactant results. Further decreases in IFT at higher concentrations of CTAB (up to 0.9 mN/m) are consistent with earlier studies indicating that saturation of the oil–water interface by surfactant molecules stabilizes at concentrations above the CMC69.
In comparison, SDS showed a similar trend, reducing IFT to 1.4 mN/m at its CMC (8 mM), which is slightly higher than CTAB but comparable to results reported by researchers in their studies of anionic surfactants in high-salinity brines. The differences in IFT behavior between CTAB and SDS reflect the variations in ionic head interactions and their compatibility with saline environments, a trend well-documented in surfactant chemistry70.
Figure 7 highlights how both surfactants outperform distilled water and brine (IFT values of 43 and 45 mN/m, respectively), which underscores the essential role of surfactants in achieving low IFT values critical for EOR applications. Comparative studies suggest that the combined effects of reduced IFT and wettability alteration, as demonstrated in this study, are pivotal for maximizing oil recovery.
Zeta potential measurement
The zeta potential results presented in Table 3 highlight the interaction between surfactants and clay particles, offering critical insights into fine migration control mechanisms. In the presence of CTAB, the positive zeta potential (+ 12 mV) indicates strong adsorption of the cationic surfactant onto clay surfaces, altering their charge. The zeta potential values were measured using a precision electrokinetic analyzer. Repeated measurements (n = 3) were conducted to ensure consistency, yielding mean values of + 12 mV for CTAB, − 43 mV for SDS, and approximately 0 mV for high-salinity brine. The measurement uncertainty was determined to be ± 2 mV, considering instrumental precision and sample preparation variations. This charge reversal promotes adhesion between clay particles and negatively charged sandstone surfaces, stabilizing particles and reducing mobility. As a result, the electrostatic attraction prevents fines from detaching and migrating through pore networks, ensuring sustained permeability during fluid displacement processes. These findings underscore the importance of CTAB in controlling fine migration through surface charge regulation, which can mitigate pore blocking and enhance oil recovery efficiency.
In contrast, the negative zeta potential (− 43 mV) observed with SDS emphasizes its distinct physicochemical behavior. The anionic surfactant increases repulsive forces between clay particles and sandstone surfaces, leading to particle dispersion. This high repulsion promotes fine movement, increasing the risk of pore blockage and reduced permeability. Similarly, the negative zeta potential of distilled water (− 40 mV) reflects an environment without stabilizing ions or surfactants, further exacerbating fine dispersion and migration. Brine, with a zero zeta potential, achieves charge neutralization through salt ions, balancing electrostatic forces and reducing fine migration risks by immobilizing clay particles. These results clearly establish that zeta potential serves as a key parameter for controlling fine migration, with CTAB proving highly effective in stabilizing particles and mitigating associated risks. The relationship between zeta potential shifts and fine migration dynamics highlights the necessity of surfactant optimization in reservoir management and EOR processes.
Contact angle measurement
Analysis of the wetting behavior of rocks under different conditions can provide a window to better understand their performance in oil recovery. In this study, the contact angles of rocks in the presence of oil and different phases, including distilled water, saline water, CTAB, and SDS solutions, were tested both in the initial conditions and after long-term training (aging) processes.
As illustrated in Fig. 8, in the initial condition, rocks in the presence of oil and distilled water, saline water, CTAB, and SDS had contact angles of 70°, 68°, 79°, and 73°, respectively. These numbers indicate the relative tendency of rocks to be wetted by water, especially when exposed to distilled water and saline water. The presence of surfactants slightly increased the contact angle, which may be due to minor changes in the surface properties of the rock, indicating a shift towards greater hydrophobicity.

The contact angle of oil droplets on (A) original surface with CTAB bulk phase, (B) oil-aged surface with CTAB bulk phase, (C) CTAB re-aged and bulk phase, (D) original surface with SDS bulk phase, (E) oil-aged surface with SDS bulk phase, (F) SDS re-aged surface and bulk phase.
After 28 days of exposure to specific temperature and pressure conditions (95°C and 2000 psi) with oil, the contact angles increased to 131°, 135°, 129°, and 132°. This indicates that the rocks have changed their surface properties due to continuous contact with oil and have become more prone to water non-wetting. The increase in contact angle indicates that the rock surfaces have become more sensitive to oil absorption, which may be due to the absorption of organic materials from the oil, which makes the surface more hydrophobic.
With the start of the re-aging process and the rocks being re-exposed in distilled water, saline water, and SDS and CTAB solutions, the contact angle values decreased to 123, 125, 91, and 93°, respectively. The re-aging process with distilled and saline water slightly decreased the contact angle but maintained a tendency towards greater hydrophobicity. On the other hand, SDS and CTAB surfactants had a more pronounced effect and were able to reduce the contact angle significantly. This reduction indicates the restoration of the hydrophilicity of the rock surface by using surfactants, which can contribute to better permeability and redistribution of water in the rocks.
These results indicate that surfactants can play an essential role in restoring or regulating the wettability of rocks, which is vital in managing oil reservoir systems. From a practical point of view, such solutions can lead to improved extraction processes and optimized use of underground resources. Using scientific and engineering methods, precise control of surface wettability significantly improves oil recovery and reduces operating costs.
Core flooding results
The results of Core Flooding experiments using clay-rich sandstone cores and under different injection conditions of CTAB, SDS, and brine solutions indicate the different effects of these solutions on permeability and RF. These experiments provide important information about the liquid phase’s behavior, the rocks’ surface properties, and the chemical interactions between the rock particles and the injected solutions. Generally, three types of solutions with different characteristics, CTAB (cationic surfactant), SDS (anionic surfactant), and brine, were injected into sandstone cores with 122 mD permeability and irreducible water saturation. Specifications and characteristics related to all 5 core samples used in this study are presented in Table 4, obtained from the initial brine flooding stage. The overall results indicate significant changes in permeability and RF caused by chemical changes on the rock surface and the physical behavior of the injected solutions.
Core flooding experiments were performed under controlled conditions (95 °C, 2000 psi). Permeability was calculated from pressure differentials and flow rates, with an uncertainty of ± 3 md, based on precision limitations of the pressure transducers and flow control systems.
Changes in the permeability of sandstone cores were measured after the injection of different solutions at a rate of 0.1 mL/min at 3 PV. These changes indicate the effect of each of the injected solutions on the permeability characteristics of the rocks. RF was also measured for each of the solutions. These values clearly show how each solution affected the oil recovery efficiency.
First core flooding
Core number 1 was flooded by CTAB solution for three PV. The RF and differential pressure data was recorded versus injection volume, as plotted in Fig. 9. After injection of the CTAB solution, the permeability decreased to 65 mD. This process leads to the creation of electrostatic forces between the clay and sand particles, which reduces the movement of fine particles (fine migration) and prevents pore blockage. This phenomenon increases rock stability and improves fluid flow inside the rock.
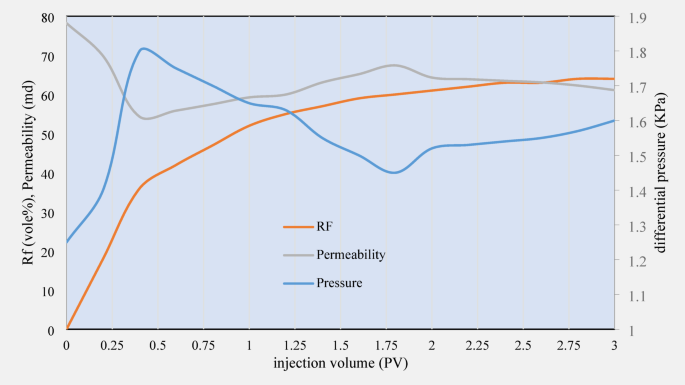
RF, differential pressure, and permeability data obtained from the first core flooding experiment.
Ions on the surface of clay particles replace the cationic ions in CTAB. This process creates electrostatic forces between clay and sand particles, which reduces fine particle movement (fine migration) and prevents pore blockage. This phenomenon increases rock stability and improves fluid flow inside the rock.
After injection into the cores, the CTAB solution recovered 65% of the oil. This result indicates the high effectiveness of CTAB in improving fluid penetration and oil recovery. The excellent performance of CTAB may be due to its ability to create a cation exchange mechanism and reduce the movement of fine particles inside the rock, which increases the efficiency of the oil recovery process. Furthermore, due to IFT reduction and wettability alteration, both of which was favorable according to previous experiments, the recovery factor reached to a very ideal value.
Core flooding 2
In this experiment, core number 2 was flooded for 3 PV with SDS solution, and the obtained results are illustrated in Fig. 10. Compared to CTAB, the permeability of sandstone cores showed a more severe decrease after injection of 8 mM SDS solution. SDS is an anionic surfactant that can cause more changes in the surface structure of rocks and is likely to cause more blockage of rock pores. This may be due to more severe changes in the rock surface composition and physical and chemical reactions occurring in contact with SDS, which cause a sharp decrease in permeability to 4 md.
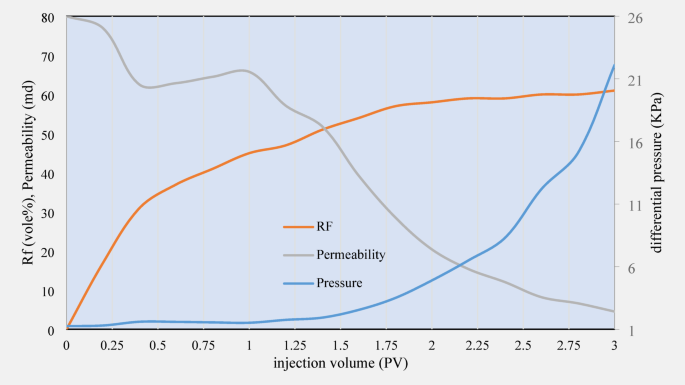
RF, differential pressure, and permeability data related to the second core flooding experiment.
The oil RF in the cores injected with SDS reached 61%, significantly lower than that of CTAB. The decrease in RF in the presence of SDS may be due to more blockage of pores and a decrease in permeability due to chemical changes of the rock surface in contact with anionic surfactants. This may be due to the accumulation of chemicals on the rock surface and the blockage of the pores or porous networks of the rock, which leads to a decrease in oil recovery efficiency.
Core flooding 3
In this experiment, core number 3 was flooded by brine for 3 PV, and the results are presented in Fig. 11. Injection of brine resulted in a relative decrease in permeability of 47 mD. Saltwater can affect the electrostatic interactions between clay and sand particles by changing the surface charge of clay particles and increasing the ionic density in the solution. This phenomenon may effectively reduce permeability, but its effect will not be as significant as that of surfactants.
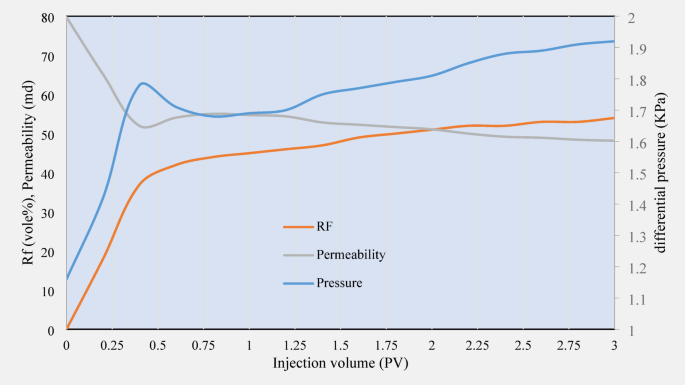
RF, differential pressure, and permeability data related to the third core flooding experiment.
The RF in cores injected with brine was 53%, the lowest of the three solutions. This value indicates that brine, despite its ability to change the surface charge of the rock, cannot recover oil and surfactants. This could be because saltwater cannot make the necessary chemical and physical changes to reduce hydrophobicity and improve the wettability of the rocks effectively enough.
The results of the Core Flooding experiments indicate that surfactants, especially CTAB, perform better in improving oil recovery and increasing permeability. The cation exchange mechanism of CTAB improves the surface properties of the rocks and reduces the movement of fine particles, which is one of the significant problems in oil recovery processes. This improvement in reducing the constriction of pores and facilitating the fluid flow increases RF.
In contrast, SDS led to a more significant decrease in permeability and RF due to more outstanding chemical and physical changes on the rock surface and pore blockage. This may be due to the more complex reactions that the anionic surfactant induces on the clayey rocks, leading to a significant decrease in oil recovery efficiency.
Finally, despite its limited effect on permeability, brine showed a poorer performance in oil RF, most likely due to its lower ability to change the surface properties of the rocks and better stimulate the movement of fluids.
Core flooding 4 and 5
Next, Core Flooding experiments conducted on two sandstone cores with irreducible water saturation showed that under different flooding conditions and injection of surfactant solutions with different concentrations and sequences, there was a significant impact on permeability characteristics and RF. These experiments were designed to investigate how the combination of surfactants affects oil recovery processes in reservoir rocks. In the first scenario (experiment 5), 2 PV of brine were injected into the core. This saline water injection typically leads to changes in fluid flow patterns and a decrease in rock permeability. Due to electrostatic effects, saline water can mobilize fine particles in the rock and reduce the free movement of fluids in the pores of the rock. Then, a 1 mM CTAB (cationic surfactant) was injected into the core. This surfactant effectively interacts with ions on the rock surface and causes cation exchange. This mechanism regulates the electrostatic forces on the rock surface, especially in the presence of fine particles or clay, ultimately leading to reduced pore blockage and improved rock permeability.
Next, an 8 mM solution of SDS (anionic surfactant) was injected into the core. The anionic surfactant SDS, which can reduce the surface tension of liquids, helps liquids penetrate better into the rock. Combining these two surfactants in a specific order and concentration improved permeability and ultimately increased the oil RF in this scenario. As a result, the oil RF in this scenario reached 81%. Ultimate data for core flooding 4 and 5 are illustrated in Table 5.
Detailed and graphical illustration of core flooding experiments number 4 and 5 is presented in Fig. 12.
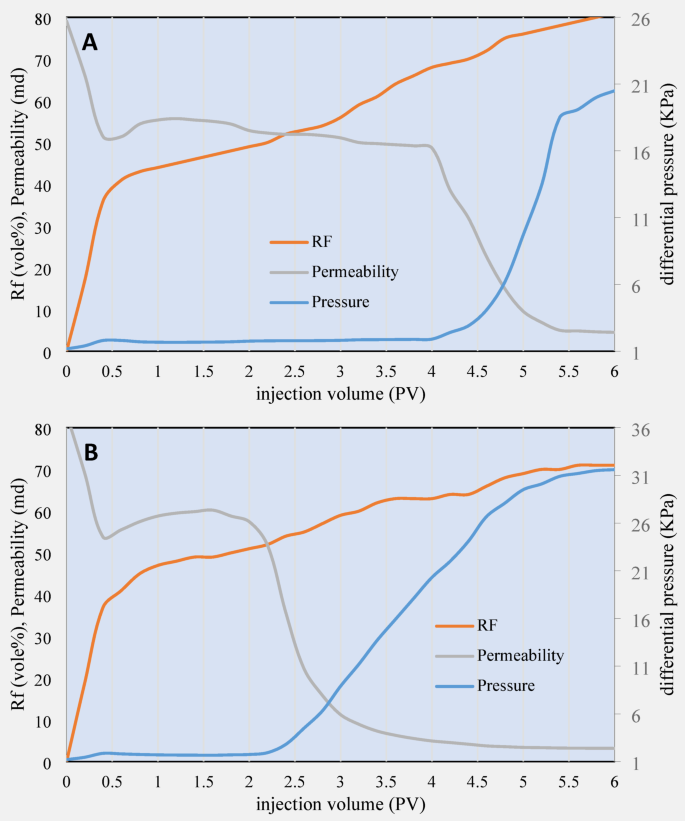
Coreflooding test results for cores number (A) 4 and (B) 5.
In the second scenario (core flooding 5), the order of surfactant injection was changed. First, 2 PV of brine were injected into the core, similar to the first scenario. However, an 8 mM solution of SDS was injected first, and then a 1 mM solution of CTAB was added to the core. This change in the injection sequence significantly impacted the oil recovery process. Since SDS, as an anionic surfactant, is injected first, its chemical and physical changes are carried out immediately, but this is done without surfactant. The previous cationic net (which creates the cation exchange mechanism) is performed, and therefore, its positive effects will not be as significant as in the first scenario. In the next step, a CTAB solution is injected, which can overcome some of the problems caused by SDS. However, because CTAB is injected as a cationic surfactant after SDS, its effect on regulating electrostatic forces and reducing pore blockage will not be as effective as in the first scenario, where CTAB was injected first. As a result, the RF in this scenario decreased to 74%. The results of these two scenarios clearly show the effect of the injection order and the concentration of surfactants on the oil recovery process. In the first scenario, where the cationic surfactant CTAB was injected before SDS, the oil recovery coefficient reached 81%. This result shows that the order of surfactant injection is of great importance because CTAB was able to play an influential role in reducing the blockage of rock pores first, and then SDS reduced the surface tension, which helped improve permeability. In the second scenario, where the order of surfactants was changed, and SDS was injected as the first surfactant, the oil recovery coefficient decreased (74%), indicating the importance of the correct interaction between surfactants and their injection order in the oil recovery process.
As compared to similar studies71 in the literature, the increase in recovery factor achieved under the proposed experimental scenarios in this research not only demonstrated a higher magnitude of recovery augmentation but also fell within the acceptable range of performance improvement for EOR techniques.
It should also be noted that the concentration of surfactants plays an essential role in determining the oil recovery efficiency. In the first scenario, where the concentration of CTAB was higher than SDS, the positive effects of CTAB in regulating electrostatic forces and improving rock permeability were more noticeable. In the second scenario, where SDS was injected at a higher concentration, the improving effects of SDS were less compared to CTAB, which led to a decrease in the oil recovery coefficient. Figure 13 best describes the mechanism of action for these aqueous phases in regard of fine migration.
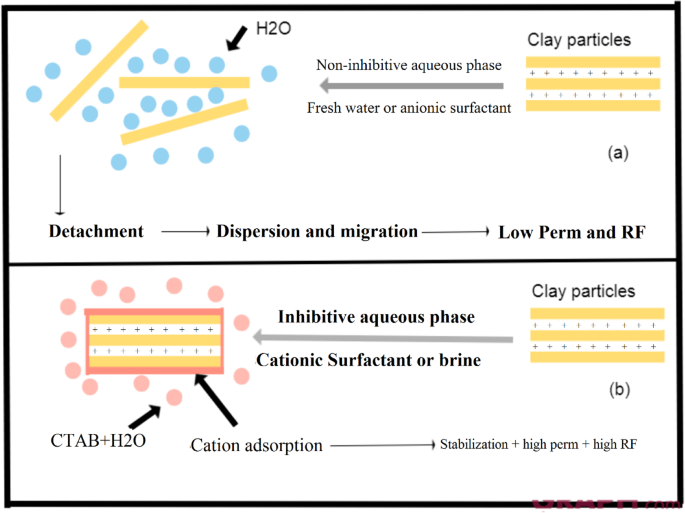
Mechanism explaining clay behavior in various aqueous phases.
link





.jpg)