Effect of phenolic anchor groups on enzymatic polymerization of coniferyl alcohol at cellulosic interfaces
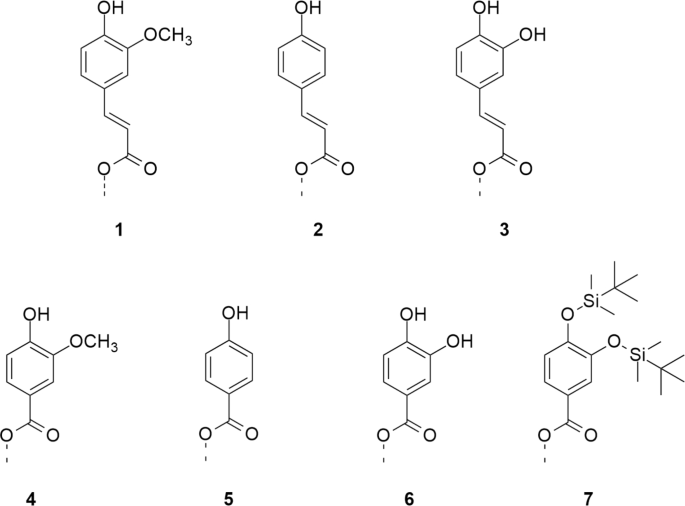
Silylation of phenolic acid esters of cellulose
For preparation of cellulosic thin films by spin coating, soluble polymers are essential. In our previous work, dissolution of cellulose ferulate in ethyl acetate was achieved via silylation28. Trimethylsilyl (TMS) groups can be cleaved off easily after spin coating to obtain cellulosic films possessing phenolic anchor groups.
In this work, cellulose esters with phenolic moieties 1-6 (Fig. 1) were synthesized according to the literature31 and subsequently converted with hexamethyldisilazane (HMDS) in the presence of catalytic TMS chloride (Fig. 2). Degree of substitution (DS) values of functional groups were determined by combining NMR spectroscopy and gravimetry. Ratios between TMS- and phenolic groups were determined by integrating the peak intensities of 1H NMR spectra. This information was included when calculating the \({\textrm{DS}_\textrm{TMS}}\) from the \({\textrm{SiO}_{2}}\) content, obtained by incineration of decomposed cellulose derivatives. The described method for determination of DS values is more exact than a previous calculation, which was based on the DS of phenolic acid ester (ferulate) prior to silylation28. In this way, errors arising from changes in DS during the reaction, by ester cleavage or accumulation in insoluble residues, could be avoided.
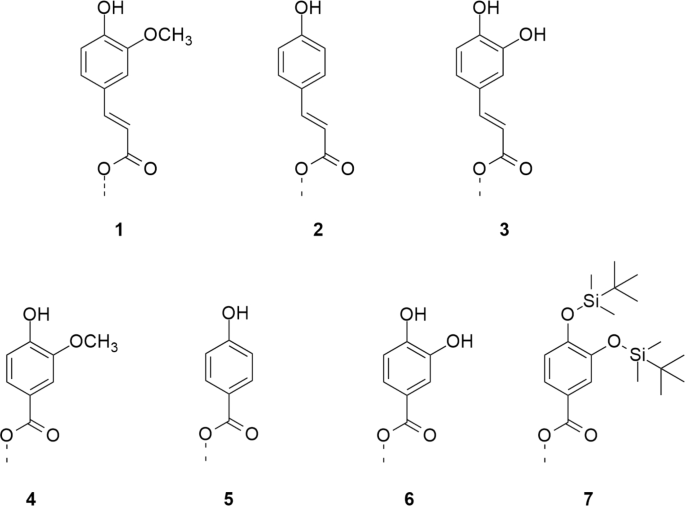
Overview of the phenolic moieties of cellulose esters serving as anchor groups for artificial lignification.
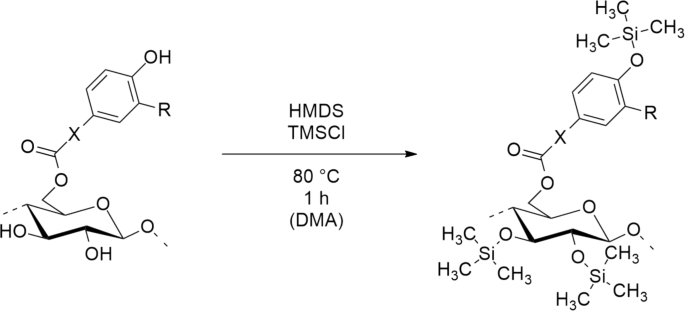
Reaction scheme for silylation of phenolic acid esters of cellulose to enable spin coating of polymers from ethyl acetate.
As shown in Table 1, \(\textrm{DS}_\textrm{TMS}\) is significantly lower for catecholic structures, i.e. caffeate (3, \(\textrm{DS}_\textrm{TMS}\) 1.72) and protocatechuate (6, \(\textrm{DS}_\textrm{TMS}\) 1.87), than for other phenolic esters (\(\textrm{DS}_\textrm{TMS}\) 2.2-2.5). Two adjacent phenolic hydroxyl groups seem to reduce the silylation reaction, which could be caused by intramolecular hydrogen bonds32,33. The values are also in line with FTIR spectra showing an increased OH vibration around 3400 to 3500 cm-1 compared to monohydroxy phenols 1,2,4, and 5 (Figure S1). However, this difference is not an obstacle for the subsequent application of regenerated cellulosic films.
Formation of model films
Cellulosic thin films with phenolic anchor groups (1-6) were obtained by spin-coating from ethyl acetate, followed by removal of silyl groups with HCl vapor. Coating parameters were adapted from literature28 to create uniform films for artificial lignification.
AFM images of spin-coated films revealed a uniform surface morphology with tiny holes, which might have arisen from solvent evaporation (Figure S2, A). However, after deprotection, films became smoother without visible flaws (Figure S2, B). The root mean square (rms) roughness of the cellulose ferulate film decreased from 2.8 nm to 1.7 nm.
Water contact angle (CA) measurements indicate similar hydrophobicities (91°-97°) for samples 1-6 arsing from TMS groups (Table 2). The influence of the type and DS value of phenolic moieties seems to be negligible. However, lower \(\textrm{DS}_\textrm{TMS}\) value of sample 6 possessing the catecholic pattern lead to less hydrophobicity, i.e. the lowest water CA of 91.5°. FTIR spectra of thin films (Figure S3) show an increased OH signal for caffeate and protocatechuate as already observed for the bulk material (Sect. 3.1).
Successful formation of model films, i.e. complete cleavage of TMS groups, is indicated by disappearance of the Si-C vibration at 1250 cm-1 and a pronounced OH signal at 3400 -1. C=O and C=C vibrations of phenolic acid esters of cellulose are still present (Figure S4).
Lignification on planar surfaces
Enzymatic polymerization of monolignols on model films in a QCM-D instrument revealed novel insights regarding lignification kinetics. Following our reproducible approach28,29, horseradish peroxidase (HRP) was adsorbed on cellulosic model films possessing phenolic anchor groups in the first step (Fig. 3). Loosely bound HRP was rinsed off with water. The frequency shift between two baselines indicated the amount of adsorbed HRP semiquantitatively and was marked as \(\Delta\)F (40). The experiment continued with introduction of coniferyl alcohol and \({\textrm{H}_2\textrm{O}_{2}}\), which resulted in a remarkable initial slope depending on the reaction speed. The amount of artificial lignin deposited could be evaluated by the difference in \(\Delta\)F and \(\Delta\)D at 40 and 110 min.
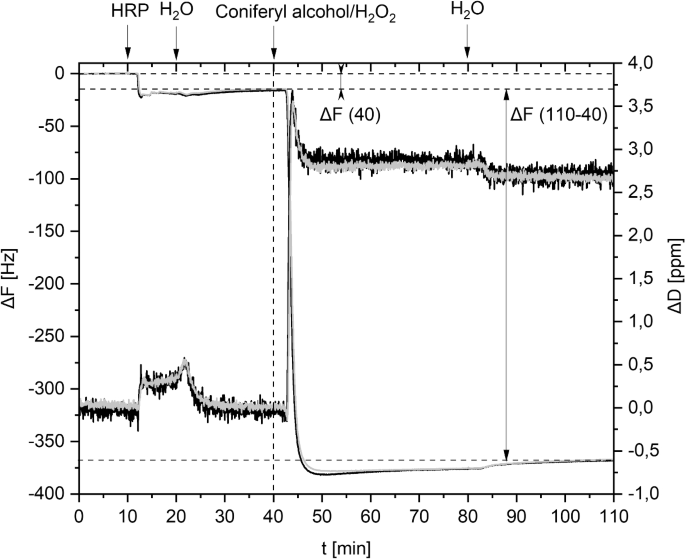
QCM-D online monitoring (\(\Delta\)F and \(\Delta\)D values over time) during enzymatic polymerization of coniferyl alcohol, example 6 (DHP@protocatechuate). Double determination is marked black and grey.
Comparing the anchor groups 1-6, there are remarkable differences in HRP adsorption (Fig. 4A). Cellulose protocatechuate (6) performed the strongest adsorption of HRP compared to surfaces 1-5. Protein adsorption is often increased by hydrophobicity of the surface. However, CAs of deprotected films (1-6) show no big differences, which could have explained a pronounced adsorption. Thus, adsorption was not only increased by hydrophobicity, but could be related to catecholic structures (3 and 6) and DS values.
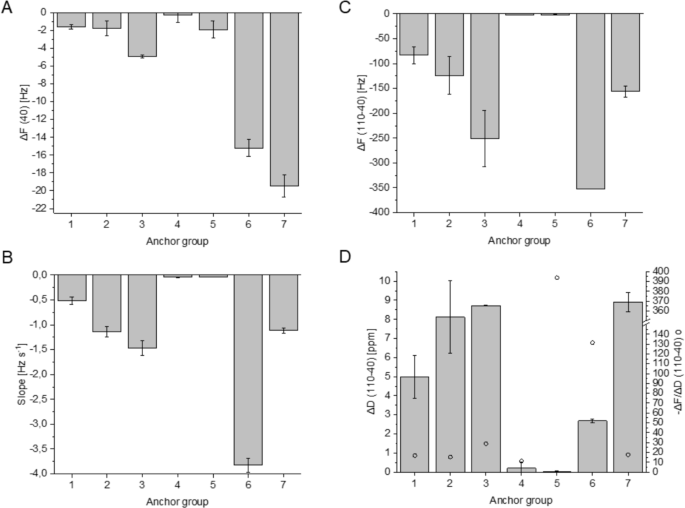
Results of QCM-D experiments dependent on anchor groups 1-7. (A): HRP adsorption (semiquantitative), (B): Initial slopes indicating velocity, (C,D): Difference of \(\Delta\)F and \(\Delta\)D at 40 and 110 min indicating amount and viscoelastic behaviour of deposited lignin.
To investigate the effects arising from hydrophobicity, cellulose protocatechuate partially protected with tert-butyl(dimethyl)silyl (TBS) groups (Fig. 2) (7) was used in reference experiments. This very hydrophobic surface (water CA 102°) led to highest HRP adsorption. However, the polymerization speed was not only dependent on the amount of HRP.
The initial slope of the \(\Delta\)F graph illustrates the speed of mass increase on surface and thus, the polymerization velocity (Fig. 4B). Slope was -3.8 Hz s-1 for protocatechuate groups (6), which showed by far the fastest process. Slope of reference 7 (-1.1 Hz s-1) is just one quarter of this value, but the amount of adsorbed HRP was higher on this surface (7). This example clearly shows the influence of anchor groups on polymerization.
It should be noted that the surface morphology of film 7 was different from that of samples 1-6. Deep holes with a diameter of up to 500 nm could be observed (Fig. S2, C). There seemed to be distinct wetting issues of the hydrophobic TBS derivative on surface during spin-coating from butanone. It could be assumed that bare silicon or gold was on the ground level of the holes. DHP deposited on film 7 possessed typical particulate structure, but there were empty areas showing size of the holes (Fig. S2, D).
Considering hydroxycinnamates, lignification process accelerated in the order ferulate (1) < p-coumarate (2) < caffeate (3). Vanillate (4) and p-hydroxybenzoate (5) performed only very slow reactions (-0.04 Hz s-1). The different behaviour of anchor groups 1 and 2 could be explained by their radical stability, as indicated by redox potential (\(\textrm{E}_\textrm{p}\)). The values for ferulic acid and its esters are in the range of +400 mV, but \(\textrm{E}_\textrm{p}\) is +736 mV for p-coumaric acid34. Owing to the low radical stability of p-coumarates, they transfer electrons to other phenols. p-Coumarates accelerate the lignification process in radical transfer reactions, but they are not incorporated integrally into the polymer35. In plants, sinapyl alcohol can be dehydrogenated in the presence of catalytic p-coumarate moieties36,37,38. Otherwise, it is a very poor substrate for peroxidases. Moreover, p-coumarate monolignol conjugates are probably controlling the size and 3D structure of lignin39.
In the case of caffeate (3), the explanation for a faster lignification is different, since \(\textrm{E}_\textrm{p}\) is +370 mV for ethyl ferulate, but +170 mV for ethyl caffeate34. High radical stability of the catecholic motif results from a resonance stabilization of the phenoxyl radical intermediate with subsequent ortho-quinone formation. However, catechol moieties are involved in the formation of benzodioxane structues in lignin.
The O-radical reacts with the favored \(\beta\)-position followed by internal trapping of the quinone methide with the adjacent phenolic OH group to the \(\alpha\)-position under rearomatization (Fig. 5). This principle was described for the incorporation of 5-hydroxyconiferyl alcohol into lignin of O-methyltransferase-deficient poplars40 and Arabidopsis41,42. In contrast to conventional \(\beta\)-O-4 linkages, rotation is blocked, and structures of the macromolecules are less flexible. Moreover, a homopolymer of caffeyl alcohol, so-called C-lignin, was found in vanilla orchid43. According to the same mechanism, benzodioxane formation occurs and is kinetically controlled, independent of enzymes or other proteins. This fast trapping step seems to be the reason why caffeate groups (3) show the best lignification performance among the three hydroxycinnamates.
The very high reaction velocity at protocatechuate groups (6) is in line with the electrochemical properties of quinones, which are excellent redox-mediators in biology and electrochemistry44,45,46. Hydroxybenzoates 4 and 5 perform only poor dehydrogenation.
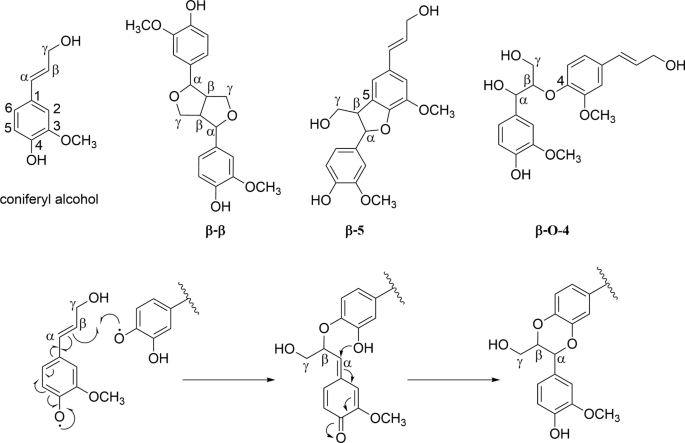
Chemical structures of coniferyl alcohol and its dimers possessing \(\beta\)–\(\beta\), \(\beta\)-5, and \(\beta\)-O-4 linkages (top). Reaction mechanism for the benzodioxane formation from coniferyl alcohol and catecholic structures (bottom).
Polymerization velocity also correlated with the final mass deposited on the films. \(\Delta\)F (110-40) was -83 Hz, -124 Hz, and -251 Hz for 1, 2, and 3 respectively (Fig. 4C). However, there were significant differences in viscoelastic behaviour. DHP@protocatechuate (6) can be considered as very rigid (Fig. 4D, \(\Delta\)F/\(\Delta\)D 131) indicating tight bonds between cellulose film and DHP particles. On the other hand, the film of reference 7 was viscoelastic (\(\Delta\)F/\(\Delta\)D 17.5), which could be explained by the bulky TBS groups on the film. Thus, formation of covalent bonds between phenolic groups and lignin was more disabled. Anchor groups possessing catecholic structures (3 and 6) were rigid (\(\Delta\)F/\(\Delta\)D>25), which suggest an effective covalent bond formation of stiff structures.
Water CA was dependent on the coverage of cellulosic films with lignin particles. On films with vanillate and p-hydroxybenzoate groups, there was almost no deposition of DHPs. These films were hydrophilic, similar to pure cellulose layers (CA 30-35°). On the other hand, high deposition rates could increase the CA up to 61°(Table 2) (6).
Molecular structure of artificial lignins
To elucidate linkages in artificial lignins formed in the presence of phenolic anchor groups, HSQC NMR measurements were conducted (Figure S5). Synthesis of sufficient amounts of dehydrogenation polymer was carried out by flow through experiments with dispersed material on a suction filter. DHPs were isolated by alkaline cleavage from the cellulose backbone and extracted with acetone29.
1H and 13C NMR resonances were assigned by consideration of HSQC NMR spectra of DHPs from the literature12 and data of dimers of coniferyl alcohol (Fig. 5) from the NMR database of lignin and cell wall model compounds47. Chemical shifts of position \(\alpha\), \(\beta\), and \(\gamma\) could be assigned to the linkage types \(\beta\)–\(\beta\), \(\beta\)-5, and \(\beta\)-O-4 as well as the \(\beta\)-O-4 benzodioxane motive.
Cross-peaks at 5.5/88 ppm and 4.8/73 ppm arising from position \(\alpha\) of the coniferyl units were integrated to determine the percentage of \(\beta\)-5 and \(\beta\)-O-4 linkages. For DHP formed at cellulose caffeate, the benzodioxane motif could be distinguished at 4.9/77 ppm. \(\beta\)–\(\beta\) bonds were quantified using the signal of position \(\beta\) at 3.0/54 ppm.
All DHP samples possessed approximately 50 % \(\beta\)–\(\beta\) linkages (Table 3). Ferulate (1) and coumarate (2) moieties lead to similar percentages of \(\beta\)-5 (\(\approx\)30 %) and \(\beta\)-O-4 (16 %) bonds. Lignification on cellulose caffeate (3) yielded DHP with low \(\beta\)-5 content (22 %) and many \(\beta\)-O-4 linkages. As assumed during QCM-D experiments (Sect. 3.3), the catechol structure was incorporated into lignin by the formation of the benzodioxane motif, which constitutes 9 % of the 32 % \(\beta\)-O-4 bonds. Protocatechuate (6) performed as an excellent redox mediator, but no benzodioxane structure could be observed. However, \(\beta\)-O-4 content was high (24 %). No DHP could be isolated from cellulose vanillate or without a matrix. DHP formed with p-hydroxybenzoate (5) consists of high amounts of \(\beta\)-5 linkages (40 %) and fewer \(\beta\)-O-4 bonds (8 %).
In general, DHPs obtained by our flow through experiments showed typical structures of Zulauf (ZL) lignin. The content of \(\beta\)–\(\beta\) linkages was very high, but 5-5 and \(\beta\)-1 could not be observed in the NMR spectra. This could be explained by high concentration of monomer radicals leading to a pronounced monomer-monomer coupling and less oligomer-oligomer coupling48.
In context with the use of DHPs as synthetic model of lignin, a similar chemical structure compared to native lignin is aimed12. For example, DHPs obtained from coniferyl alcohol possess higher \(\beta\)-5 and \(\beta\)–\(\beta\) contents but lower amount of \(\beta\)-O-4 bonds than natural lignin. Thus, a pronounced increase of \(\beta\)-O-4 linkages is desired and could be achieved by phenolic groups 3 and 6. The anchor group caffeate increased the \(\beta\)-O-4 content by the formation of benzodioxane structures. Moreover, high deposition rates provide hydrophobic DHP aggregates, which should be beneficial for \(\beta\)-O-4 bonding12. In particular, this could be the effect of protocatechuate groups, which did not lead to benzodioxane structures but rather high \(\beta\)-O-4 content. On the other hand, anchor groups with poor performance lead to very low \(\beta\)-O-4 content (5) or even no aggregation of DHP (4).
Py-GC-MS chromatograms (Figure S6) of DHPs show specific peaks arising from the anchor groups incorporated into the lignin structure (Figure S7). The signals of ferulic acid, p-coumaric acid, and p-hydroxybenzoic acid are clearly visible. For the caffeate structure, the fragments catechol, methyl catechol, and ethyl catechol are visible. Catechol and methyl catechol were also detected during pyrolysis of protocatechuate moities. Moreover, pyrolysis of p-coumaric acid leads to an intense signal of coumaran. It could be assumed, that p-coumaric moieties are less covalently linked to lignin than e.g. ferulic acid, which is comparable to the situation in natural plants (Sect. 3.3).
The molecular mass distribution of DHPs obtained with hydroxycinnamates correlates with the reaction velocities and the deposited amounts observed in QCM-D experiments (1<2<3) from ferulate (\(\textrm{M}_\textrm{w}\) 1550) to p-coumarate (\(\textrm{M}_\textrm{w}\) 5501) to caffeate (\(\textrm{M}_\textrm{w}\) 6902, Table 3). However, molar masses of DHPs from hydroxybenzoates (5-6) are lower in general, and there seems to be no correlation with the lignification speed or deposited mass.
link





.jpg)