Design of a combined LED and rapid-injection NMR system for structure elucidations and kinetic analyses
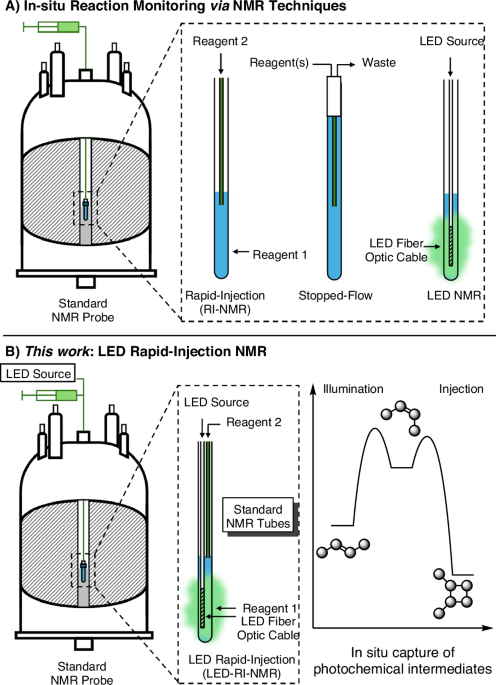
Gaining inspiration from the McGarrity, Ogle, Reich, and Denmark RI-NMR systems, our approach was focused on developing an apparatus that did not require modifications to the NMR spectrometer, probe, or facility2,22. We describe below an overview of the LED-RI-NMR system; however, detailed schematics are available in the Supporting Information. The LED-RI-NMR system was adapted for a Varian VNMRS 500 MHz spectrometer equipped with a 10 mm Varian NMR probe. The barrel of the injector was constructed from a hollow aluminum rod 53 cm in length equipped with a height-adjusting handle and injector tip assembly (Fig. 3). The injector tip assembly has two perforations at the bottom fitted with a 3 mm sealed capillary for the fiber optic cable and an open 1 mm capillary for RI-NMR (Fig. 3E). The fiber optic cable was fed through the aluminum rod to a Prizmatix multiple wavelength LED box capable of emitting light from four different wavelengths: (A) 402 nm, (B) 454 nm, (C) 524 nm, and (D) 631 nm (Fig. 4A–D). The injector capillary was connected to a polymer threaded coupling (1/4-28 NPT) equipped with 5.4 mm OD (0.1 mm ID) Teflon O-rings and coupled with Teflon tubing and Tefzel microfluidic ferrules and nuts to a Harvard Apparatus syringe pump. With the LED-RI-NMR system in hand our investigations switched to calibrating the delivery of LEDs, organic media, as well as mixing.

A Polyacetal height adjusting handle. B Aluminum injector barrel with Teflon spacers. C Polyacetal injector tip assembly. D Multi-channel LED box. E Close-up of Injector tip assembly with injection capillary (left) and LED capillary (right).

A 402 nm, B 454 nm, C 524 nm, D 631 nm.
Introducing light into NMR samples presents a challenge; ensuring compatibility with the LED source, associated equipment, and the NMR spectrometer is often arduous. Integrating LEDs into the NMR probe enables simultaneous illumination of various samples and enables rapid-injection experiments. However, this approach requires significant modification to the probe. While placing an LED lamp at the top of the NMR bore is an option, this raises concerns about the efficiency of sample illumination due to the large distance between the lamp and the sample. To address this issue, we sought a method to directly illuminate the sample without modifying the NMR probe or sample tube. Inspired by Gschwind’s previous LED-NMR setups11, we utilized a sanded polycarbonate fiber optic cable encased in a 3 mm sealed glass capillary. This configuration allows for direct illumination of the NMR sample, and since the fiber optic cable is part of the LED-RI-NMR assembly, no modifications to the NMR tube or probe are required for illumination experiments. Additionally, this placement of the fiber optic cable enables uniform illumination of the sample in the measuring coils of the NMR spectrometer (Fig. 5).

Dashed box indicates the location of the measuring coils for the NMR spectrometer.
A common issue in RI-NMR experiments is the injection of reagents into the sample without early detection. Since the LED-RI-NMR system remains static inside the sample while collecting spectra, it was not feasible to position the injector tip at the same depth as the LED capillary. To avoid early detection of the injected reagent, we explored the idea of raising the injection capillary above the sample. After trial and error, it was found that positioning the injector tip 4 cm above the solution in the NMR tube effectively prevented early detection while enabling successful reagent delivery. This injection system demonstrated a linear range (Fig. 6) of injected volume between 100 and 500 μL (slope = 0.9803 ± 0.016).

Injections were performed in 10 mm NMR tubes using 2 mL of THF and injecting between 100 and 500 μL of benzene into the sample.
Often the biggest challenge in RI-NMR experiments is achieving fast and efficient mixing. By submerging the LED capillary in the NMR sample and spinning the NMR tube at 6 Hz, effective mixing of Blue #1 in MeOH (Fig. 7A–D) is rapidly achieved (<1 s) with a 200 µL injection (See Supplementary Video 1). Key to success in our system was the LED-capillary, which provided a fixed mixer. With the finished injector in hand, we then sought out well-tested and reported reactions to validate the practicality of this spectroscopic tool.

A Pre-injection with 2 mL of MeOH in a 10 mm NMR tube spinning at 6 Hz. A video of the injection sequence is available as Supplementary Video 1. Injection at B 0.55 s, C 1.29 s, and D 3.02 s.
To calibrate the LED-RI-NMR system for mechanistic investigations, we needed to demonstrate its ability to mimic standard bench reaction protocols that require illumination as well as the addition of reagents over a range of temperatures and time frames under inert atmospheres. Of note, the illumination and injection features were designed to be independent from one another and compatible with VT-cooling and heating. Therefore, our efforts became focused on testing: (1) the illumination feature (without rapid injection) by exploring a photocatalytic reaction that has all of the reaction components pre-mixed; (2) the illumination feature in tandem with rapid injection to photogenerate an intermediate and capture it by rapid injection; and (3) the illumination feature in concert with rapid injection to generate and investigate an intermediate under constant irradiation.
The construction of the LED-RI-NMR injector system was initiated in part by our recent interest in photoredox-enabled reactions, wherein we opted to study the photosensitized generation of carbonyl ylides. Carbonyl ylides are a type of 1,3-dipole generated by a variety of methods that react readily with electron-deficient alkenes to form highly substituted tetrahydrofurans23. Recently, strategies to generate carbonyl ylides through redox-neutral photocatalysts have allowed for the synthesis of various other small molecules24,25. While the direct photolysis of epoxides to carbonyl ylides have been studied, kinetic studies on the photosensitized reactions are scarce and rely on laser flash photolysis instead of bulk solution monitoring26,27.
We began by combining trans-stilbene oxide 10 with citraconic anhydride 11 in benzene-d6 with the acridinium photocatalyst 12. The 1H NMR spectra were recorded over a course of 22 h under constant irradiation at 454 nm allowing for the activation of the photocatalyst 12, decay of starting materials 10 and 11, as well as the formation of product 13 to be monitored (Fig. 8A). The initiation of 12 into its photoactive state was completed within minutes (t½ = 11.27 ± 0.19 min) following a first-order kinetic model under constant light intensity28. Due to the paramagnetic nature of the active photocatalysts, these intermediates could not be detected by 1H NMR spectroscopy. Of note, the C-C opening of epoxides using photosensitizers is traditionally performed with cyanoarenes as photocatalysts; however, we found that acridinium photocatalysts, such as 12, are compatible in the desired reaction19.

A Reactions were conducted using 2 mL of dry, degassed benzene-d6 in a 10 mm NMR tube. Samples were prepared inside of an argon glovebox by adding to the NMR tube trans-stilbene oxide 10 (30 μmol, 1.0 equiv.), citraconic anhydride 11 (30 μmol, 1.0 equiv.), photocatalyst 12 (3 μmol, 0.10 equiv.), and 1,1,2,2-tetrachloroethane (30 μmol) as an internal standard. B Kinetic data for the photoredox neutral [3 + 2]-cycloaddition reaction. Kinetic data are an average of duplicate runs; negative signs indicate consumption of starting material; positive signs indicate formation of products; solid lines indicate theoretical fit (first order and zeroth order) calculated using the CurveFitter toolbox in Matlab at a 95% confidence interval. Kinetic formulas and fit parameters are available in the Supplementary Information under Section 3: Experiment 1.
The kinetic order dependence in 10, 11, and 12 was then determined by monitoring the formation of 13 by 1H NMR over a range of concentrations independently. The decay of stilbene oxide 10 followed a first-order kinetic model under pseudo-steady-state conditions (kobs = 2.99 ± 0.18 × 10−5 s−1) and was found to exhibit a first order dependence on the reaction (Fig. 8B). Of note, upon varying the concentration of citraconic anhydride 11 from 13 to 43 mM the rate of the reaction was found to not be affected and is consistent with a zero-order dependence. The determination of overall rate equation was determined to be rate = kobs[10]1[11]0[12]0 (The orders of reagents were determined to be first order in 1, zero order in 2, and zero order in catalyst 3. See SI for details), which indicates that the generation of the carbonyl ylide CY is likely rate-determining (Fig. 8B). With the illumination aspect of the LED-RI-NMR system proven successful, our investigations switched to examining the injection aspect of the system (Figs. 9, 10).

A Reactions were conducted using 2 mL of dry chloroform-d in a 10 mm NMR tube. Samples were prepared by adding 3-diazo-3-phenylacetone 14 (30 μmol, 1.0 equiv.) and 1,1,2,2-tetrachloroethane (30 μmol) as an internal standard to the NMR tube and dissolved in dry CDCl3 (2 mL). The sample was illuminated with 454 nm light using the LED-RI-NMR injector system for ca. 1 h at which point all 14 was consumed. Diethylamine (30 μmol, 1.0 equiv.) was then promptly injected via LED-RI-NMR syringe pump (500 μL, 14 mL/min). Kinetic data are an average of duplicate runs; negative signs indicate consumption of starting material; positive signs indicate formation of products; solid lines indicate theoretical fit (first order and second order) calculated using the CurveFitter toolbox in Matlab at a 95% confidence interval. Kinetic formulas and fit parameters are available in the Supplementary Information under Section 3: Experiment 2 B Stacked 1H NMR array of the aliphatic region for the conversion of 14 to 15 under 454 nm illumination.

A Reactions were conducted using 2 mL of dry chloroform-d in a 10 mm NMR tube. Samples were prepared by adding trans–17 (30 µmol, 1.0 equiv.), 1,1,2,2-tetrachloroethane as an internal standard (30 µmol) to the NMR tube and dissolved in CDCl3 (2 mL). The sample was illuminated with 454 nm light using the LED-RI-NMR injector system for ca. 10 min at which point equilibrium was reached between trans–17 and cis–17. 3-diazo-3-phenylacetone 14 (30 μmol, 1.0 equiv.) was then promptly injected via LED-RI-NMR syringe pump (500 μL, 14 mL/min) with simultaneous 454 nm illumination. Kinetic data are an average of duplicate runs; negative signs indicate consumption of starting material; positive signs indicate formation of products; solid lines indicate theoretical fit (first order) calculated using the CurveFitter toolbox in Matlab at a 95% confidence interval. Kinetic formulas and fit parameters are available in the Supplementary Information under Section 3: Experiment 3. B Stacked 1H NMR array of the aromatic region of the isomerization from trans–17 to cis–17.
Although the generation of synthetic intermediates in situ is operationally simple for bench settings it often becomes logistically challenging for mechanistic investigations due to engineering a reliable delivery system that accurately transfers precise quantities of reagents at a known time and temperature. Therefore, our focus switched to investigating a Wolff rearrangement that requires illumination to generate a synthetic intermediate (ketene) followed by the injection of a subsequent reagent to explore its chemical reactivity.
The irradiation of diazoketones into ketenes and their subsequent reactions has been extensively utilized in various synthetic applications29,30. Numerous studies on the Wolff Rearrangement have provided evidence for both concerted and stepwise mechanisms for the 1,2-rearrangment following loss of dinitrogen31. The utility of ketenes produced has been exemplified in various recent total syntheses and cycloaddition strategies32,33. While the conversion of diazoketones to ketenes has been studied thermally and photochemically, the ability to subsequently investigate the reactivity of the ketene intermediate with nucleophiles is challenging because of the incompatibilities between diazoketones and nucleophiles. As a result, we became interested in these processes and saw it as an opportunity to test the rapid-injection component of the LED-RI-NMR system where we could photochemically generate a reactive ketene and subsequently investigate its capture with alcohols and amines for the synthesis of esters and amides – mimicking bench settings.
We first started with 3-diazo-3-phenylacetone 14 in dry CDCl3 and began irradiation (454 nm) at 25 °C such that the 1H NMR signals could be monitored. A first-order decay of 14 and formation of ketene 15 was observed with kobs values of (5.35 ± 0.16 × 10−4 s−1) and (4.77 ± 0.16 × 10−4 s−1), respectively, indicating that the decay of 14 and formation of 15 coincide. Of note, the photogenerated ketene 15 was stable in solution at 25 °C with no detectable change to the 1H NMR spectrum over extended periods of time (>1 h). With the ketene 15 prepared cleanly inside the NMR probe, our investigations switched to probing its chemical reactivity by injecting nucleophiles.
Initially, we sought to compare the reactivity of ketene 15 with methanol versus diethyl amine (Et2NH); however, upon injection of 1.0 equiv of MeOH into a CDCl3 solution of 15 no reaction occurred34. Even upon heating the sample to 60 °C no reaction proceeded and 15 remained unchanged. In a separate case, the injection of diethylamine induced a rapid reaction with 15 and generated the corresponding amide 16 following a second order model (kobs = 21.91 ± 0.54 M−1 s−1). Upon, varying the concentration of diethylamine from 8 mM to 20 mM an order dependence of one was observed35. In addition, ketene 15 was found to exhibit a first-order dependence on the reaction – providing the following rate equation rate = kobs[15]1[Et2NH]1.
The LED-RI-NMR system enabled us to conduct the Wolff rearrangement entirely within the NMR spectrometer, allowing for real-time monitoring of ketene 15 and enabling reagent injections to probe reactivity. At this point, we have utilized each component (LED incorporation and rapid-injection) separately; however, an advantage to the LED-RI-NMR system is the ability to use these components simultaneously to capture photochemical intermediates.
With the successful demonstration of the illumination and injection features in tandem, our investigations became focused on exploring the injection of reagents while the sample was under constant illumination. Therefore, we chose to investigate a photostationary state or light-induced pre-equilibrium, such that we could investigate the chemical reactivity of one species selectively. In recent years, the photoinduced isomerization of azobenzenes has gained considerable interest in various fields, such as photoswitches and phototherapies, making it an ideal and important system to investigate36,37,38,39.
Irradiation of trans–17 initiates the isomerization into cis–17 until equilibrium is reached; the subsequent ratios of trans-/cis–17 is dependent on the wavelength of light used40. Reactions of cis–17 that are not competitive with trans–17 are relatively rare, with only a few photoinduced examples in the literature41,42. Notably, the [2 + 2]-cycloaddition of cis–17 with ketenes has been reported, where trans–17 does is unreactive43. Given our ability to generate ketenes from diazoketones, as shown in the previous example, we saw this as a prime opportunity to study this reaction. First, we needed to determine the photostationary state of 17 and the trans-/cis– ratio following visible light illumination. A small portion of trans–17 absorbs blue light (380–500 nm), and illumination at 454 nm in CDCl3 using the LED-RI-NMR system yielded a 75:25 trans-/cis–17 ratio after approximately 10 min. Both the decay of trans–17 and formation of cis–17 followed a first-order equilibrium model (Keq = 0.25 ± 0.01) until the photostationary state was reached. Once equilibrium was reached, 14 was promptly injected into the sample with the light on. Initially, minor spectral changes were observed, but over 8 h, a clear conversion of 14 and 17 was observed into the corresponding cycloaddition product 18.
There are several points to be made about the reaction, particularly with how each reagent is involved in the production of 18. First, upon irradiation of 14 at 454 nm, the Wolff Rearrangement occurs, generating ketene 15. However, because 15 rapidly undergoes cycloaddition with cis–17, it is consumed as soon as it is generated. The decay of 14 proceeds in a first-order model (kavg = 1.14 ± 0.03 × 10−4 s−1) as shown in earlier experiments; however, due to other reagents that absorb light in the reaction medium, the rate of diazo decay is slightly diminished. Second, trans–17 decays in the reaction following a first-order model (kavg = 3.89 ± 0.12 × 10−4 s−1) akin to the consumption of cis–17 via the photostationary state between trans-/cis–17. As predicted, trans–17 does not participate in the [2 + 2]-cycloaddition but rather acts as a source of cis–17. Third, the cycloaddition product 18 is formed following a pseudo-first-order model (kavg = 4.30 ± 0.15 × 10−4 s−1). We rationalize this model to the low concentration of ketene at any given time in the sample. Finally, cis–17 follows a pseudo-first-order kinetic model (kavg = 3.32 ± 0.12 × 10−4 s−1) where it retains the same 75:25 ratio with trans–17 under equilibrium conditions.
In conclusion, we have developed an LED-RI-NMR system that can be utilized for a variety of mechanistic investigations involving light and reactive intermediates. We have designed this apparatus to be simple and practical for academic and industrial laboratories for the study of photoinduced and photocatalytic reactions, photosensitive proteins44, and for the discovery of new chemical reactions initiated by light. We have exemplified the utility of this system by studying three different photo-driven reactions that employ different aspects of the LED RI-NMR system. We foresee that this system is advantageous to both LED NMR and RI-NMR systems separately and stands on its own for new mechanistic investigations. We will continue to use this system for the development of new chemical reactions as well as develop new RI-NMR systems for the modern age of chemistry.
link





.jpg)