Scientists Find First Natural Evidence Of Chemical Reaction That May Have Made Life On Earth Possible


Signs of this chemical reaction in nature have eluded scientists for decades. (Credit: Gallwis on Shutterstock)
Life on our planet may have started via a process fueled by hydrothermal vents on the ocean floor billions of years ago, and that process continues today.
In A Nutshell
- For decades, lab experiments showed underwater volcanoes could convert nitrogen gas into ammonia without any biological help, but scientists had never found proof it actually happened in nature.
- Researchers analyzing rock cores from beneath the South China Sea have now documented the first convincing geological evidence of this reaction.
- By examining mineral-filled veins that formed deep in oceanic crust, the team found an unmistakable chemical fingerprint that only abiotic reactions could produce.
- At the production rates estimated in this study, prebiotic oceans could have accumulated enough dissolved nitrogen to support life within about 250,000 years, a geological blink of an eye.
Long confirmed in labs, scientists have finally uncovered real world evidence of underwater chemical reactions suspected to have facilitated life on Earth.
For more than 30 years, laboratory experiments have shown that underwater volcanic systems can convert nitrogen gas into ammonia, a compound essential for building proteins and DNA. The chemistry worked beautifully in beakers and pressure vessels. Yet when researchers examined actual rocks from the seafloor, the telltale signs of this reaction were nowhere to be found. The disconnect between what happened in labs and what happened in nature left a frustrating gap in understanding how early Earth could have supplied the nitrogen compounds that life requires.
Now, a study analyzing rock cores drilled from beneath the South China Sea has finally bridged that gap. Researchers report the first convincing geological evidence that this reaction, called abiotic nitrogen reduction, actually occurs in submarine hydrothermal systems. In plain terms, hot underwater volcanic vents can turn inert nitrogen gas into a form of nitrogen that living things can use, and they do it without any help from biology.
The discovery, described in Nature Communications, transforms a theoretical possibility into a documented geological process and suggests Earth may have become hospitable to life much faster than scientists once thought.
Why Nobody Could Find It Before
The search for this reaction in nature has frustrated scientists for decades, not because it doesn’t happen, but because its chemical fingerprint gets erased before anyone can detect it.
Ammonia produced deep in oceanic crust must travel upward through rock and sediment before reaching the seafloor. Along the way, these fluids mix with shallower waters that contain ammonia from biological sources: decomposing organic matter, nitrogen compounds dissolved in seawater, and material from sediments. By the time volcanic fluids vent into the ocean, any ammonia from deep chemical reactions has been diluted and overwritten by nitrogen that came from living things.
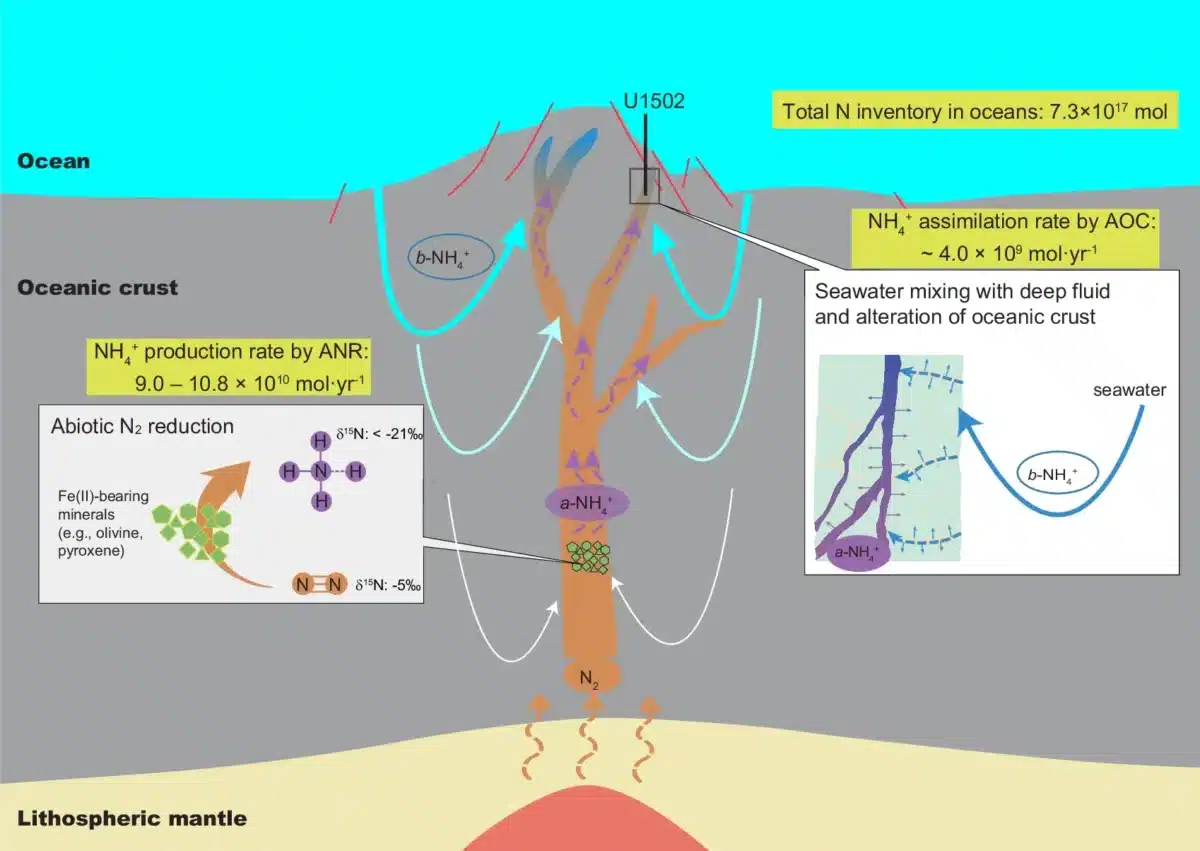
The research team, led by scientists from the Chinese Academy of Sciences and the University of Alberta, realized they needed to look somewhere else entirely. Instead of sampling fluids that had already mixed with shallow waters, they examined hydrothermal veins. These are mineral-filled cracks that formed when hot fluids flowed through fractured rock far below the seafloor. The veins act like time capsules, preserving the chemical signature of deep fluids before they could be contaminated by biological nitrogen trickling down from above.
A Telltale Chemical Signature
Rock cores from a site called Hole U1502B, drilled into seafloor crust between 16 and 32 million years old, revealed the signature the team was hunting for.
Nitrogen atoms come in two varieties: a lighter version and a heavier version. The ratio between these two forms acts like a fingerprint, revealing where nitrogen came from and what happened to it along the way. Nitrogen from biological sources, the kind that comes from decomposing sea creatures or dissolved in ocean water, carries a distinctive fingerprint that scientists can easily recognize. So does nitrogen from Earth’s deep interior.
The deepest vein samples carried a fingerprint unlike either of these sources. The nitrogen was far lighter than anything biological processes could produce, and even lighter than what comes directly from the mantle. Something was stripping away the heavy nitrogen atoms to an extreme degree.
The culprit, according to the researchers, is the chemical reaction they were searching for. When nitrogen gas encounters iron-rich minerals in hot conditions, the lighter nitrogen atoms react more readily than the heavier ones. This sorts the nitrogen by weight, concentrating light atoms in the ammonia product while leaving heavy atoms behind in the leftover gas. The extreme sorting seen in the U1502B samples matches what theory predicts for this reaction.
The pattern in the rock cores sealed the case. The deepest samples showed the lightest nitrogen and the highest concentrations. Moving toward the surface, samples became progressively heavier and contained less nitrogen. This gradient traces the boundary where deep volcanic fluids carrying abiotic ammonia mixed with shallow fluids carrying biological nitrogen from seawater, exactly what the researchers expected to find.
How Rocks Make Ammonia
The reaction relies on iron-rich minerals that are common in oceanic crust. When hot water carrying dissolved nitrogen gas encounters these minerals, the iron pulls apart the nitrogen molecules. Nitrogen gas is notoriously stable because its two atoms are locked together by an extremely strong bond, one of the strongest in nature. The iron provides the chemical muscle to break that bond. Once freed, nitrogen atoms combine with hydrogen from the surrounding water to form ammonium, a nitrogen compound that can dissolve in seawater and eventually get incorporated into living organisms.
Nitrogen gas feeds into this system continuously. Volcanic activity at mid-ocean ridges releases nitrogen from Earth’s interior, providing raw material for the reaction wherever hot fluids circulate through iron-bearing rock.
Filling an Empty Ocean
Based on their measurements, the researchers estimate that this reaction could deliver a substantial amount of ammonium to the oceans each year, roughly comparable to the total amount of nitrogen gas that volcanoes release from Earth’s interior. In today’s oceans, which already contain enormous quantities of dissolved nitrogen compounds, this contribution barely registers.
On the early Earth, the situation was completely different. Before life evolved, the oceans likely contained very little dissolved nitrogen in usable form. At the production rates estimated in this study, and accounting for nitrogen that gets absorbed back into seafloor rocks, the researchers calculate that prebiotic oceans could have accumulated enough dissolved nitrogen to match modern levels in roughly 250,000 years.
To put that timeframe in perspective, 250,000 years is to Earth’s 4.5-billion-year history what about half an hour is to a calendar year. In geological terms, the oceans could have filled with life-supporting nitrogen almost instantly.
Conditions on the early Earth would likely have made this process even more productive. The young planet’s crust contained more of the iron-rich rocks that drive the reaction. More of those rocks meant more opportunity for nitrogen gas to be converted into ammonia.

A Process Billions of Years Old
Evidence that this reaction has operated throughout Earth’s history was already sitting in the scientific literature, though its full meaning may not have been appreciated. Volcanic rocks from the Pacific Ocean floor dating back 130 to 170 million years show the same lightweight nitrogen signature. So do rocks from the Atlantic dating to 120 million years ago.
The most intriguing evidence comes from minerals that are 3.5 billion years old, formed when Earth was barely a billion years into its existence. Ammonia trapped inside tiny fluid pockets in these ancient minerals carries the telltale lightweight signature of abiotic production. This suggests the reaction was already operating when life on Earth was just getting started, potentially providing a critical nutrient source for the earliest organisms.
The failure to detect this process in modern volcanic vent fluids now makes sense. When scientists sample those fluids, they’re collecting material from the shallow, mixed zone where biological nitrogen dominates. The abiotic signal remains hidden at depth, preserved only in minerals that formed from deep fluids before mixing could occur. The researchers found it by looking in a place nobody had thought to check.
Ammonia also traps heat in the atmosphere, functioning as a greenhouse gas. The young Sun was dimmer than it is today, yet geological evidence indicates early Earth was warm enough for liquid water. Scientists have long puzzled over how the planet stayed warm under a weaker Sun. If submarine volcanic systems supplied ammonia to early oceans at the rates this study indicates, some of that ammonia could have escaped into the atmosphere and helped warm the planet.
The transition from laboratory demonstration to geological detection closes a decades-long gap between theory and observation. The chemistry that makes ammonia from volcanic heat and iron-rich rock is not merely possible in principle. The new evidence, together with older clues, suggests this chemistry has likely been active in many underwater volcanic systems for much of Earth’s history. Long before industrial factories learned to manufacture ammonia for fertilizer, the planet was doing it naturally, stocking the oceans with one of life’s essential ingredients.
Paper Summary
Limitations
The study’s estimates represent an upper bound rather than a definitive measurement of global ammonia production. The researchers acknowledge that the lower limit cannot yet be determined due to insufficient data from other locations. Some of the calculations rely on theoretical models and proxy measurements rather than direct experimental values because the relevant laboratory data do not yet exist for all the minerals involved. The samples come from a single drilling site in the South China Sea, and while similar chemical patterns have been reported from other oceanic locations, extrapolating from one site to worldwide estimates introduces uncertainty. How ammonia production rates on early Earth compared to modern rates also remains speculative, since the composition and behavior of ancient oceanic crust are incompletely understood.
Funding and Disclosures
The authors declare no competing interests. Funding was provided by the National Natural Science Foundation of China through multiple grants, the Guangdong Special Support talent team Program, the National Key R&D Program of China, the Ministry of Science and Technology of China, and an NSERC-Discovery Grant from Canada.
Publication Details
Authors: Liheng Sun, Kan Li, Zhen Sun, Yunying Zhang, and Long Li
Affiliations: South China Sea Institute of Oceanology, Chinese Academy of Sciences (Guangzhou, China); University of Alberta (Edmonton, Canada); Guangzhou Marine Geological Survey, China Geological Survey (Guangzhou, China); China-Pakistan Joint Research Center on Earth Sciences (Islamabad, Pakistan)
Journal: Nature Communications | Published: November 28, 2025 | DOI: | Citation: Sun, L., Li, K., Sun, Z., Zhang, Y. & Li, L. “Abiotic N₂ reduction in submarine hydrothermal systems could quickly fertilize prebiotic oceans,” published November 28, 2025
link





.jpg)