Materials
The following compounds and reagents were obtained from commercial suppliers and used without further purification: Chloroform-d, >99.80% (CDCl3, Apollo Scientific); N,N-Diisopropylethylamine, 99% (DIPEA, abcr GmbH); N-(9-Fluorenylmethoxycarbonyl)-l-alanine (Fmoc-Ala-OH, Novabiochem); 2-(dimethylamino)pyridine (DMAP, Fluorochem); N,N-dicyclohexylcarbodiimide, 99% (DCC, Acros Organics); piperidine, 99% (Acros Organics), N-acetyl-l-alanine (GL Biochem); hydroxy-telechelic poly(ε-caprolactone) (Mn = 10,000 g/mol specified, Mn = 22,000 g/mol according to GPC analysis) (Aldrich); tetrahydrofurane (THF), DCM, and methanol were purchased from Reactolab SA and distilled prior to use.
Synthesis of mPCL
The end-modified mPCL was prepared in four steps as described in the literature18:
PCL20-AlaFmoc (1)
Hydroxy-telechelic poly(ε-caprolactone) (Mn = 20,000, 100 g) was dissolved in THF (700 mL). Fmoc-Ala-OH (17.7 g, 113 mmol), DMAP (3.45 g, 28.2 mmol), and DCC (35.3 g, 171 mmol) were added, and the reaction mixture was stirred at room temperature for 3d. The mixture was poured into MeOH. The precipitate was dried under high vacuum to yield PCL20-AlaFmoc 1 as a colorless solid in a yield of 97 g (97%). 1H NMR (400 MHz, CDCl3): δ = 7.80 (d, 2H), 7.61 (m, 3H), 7.41 (m, 3H), 7.34 (m, 4H), 5.42 (bs, 1H), 4.40 (brs, 7H), 4.26 (brs, 100H), 4.08 (t, 198H), 2.32 (t, 200H), 1.68 (m, 426H), 1.41 (m, 213H) ppm.
PCL20-AlaNH2 (2)
PCL20-AlaFmoc 1 (95 g) was dissolved in DCM (400 mL), and piperidine (5.0 g) was added. The reaction mixture was stirred at room temperature for 1 d and then poured into MeOH (1.5 L). The precipitate was re-dissolved in DCM and poured again into MeOH. The precipitation procedure was repeated one more time. The precipitate was washed with MeOH and hexane and was dried under high vacuum to yield PCL20-AlaNH2 2 as a colorless solid in a yield of 79 g (83%). 1H NMR (400 MHz, CDCl3): δ = 4.09 (t, 1.94H), 3.92 (t, 0.03H), 3.57 (m, 0.02H), 2.33 (t, 2.00H), 1.67 (m, 4.26H), 1.41 (m, 2.16H) ppm.
PCL20-Ala2Fmoc (3)
Fmoc-Ala-OH (7.35 g, 47.2 mmol), DIEA (3.78 g, 28.5 mmol), and PyBOP (13.2 g, 25.7 mmol) were added to a solution of PCL20-AlaNH2 2 (70 g) in THF (700 mL). The reaction mixture was stirred at room temperature for 3 d and then poured into MeOH (1.2 L). The precipitate was re-dissolved in DCM, and the solution was again precipitated into MeOH. The precipitate was collected, washed with hexane, and dried under high vacuum to yield PCL20-Ala2Fmoc 3 as a colorless solid in a yield of 70 g (99%). 1H NMR (400 MHz, CDCl3): δ = 7.79 (d, 0.03H), 7.61 (d, 0.03H), 7.42 (m, 0.03H), 7.34 (m, 0.04H), 6.46 (brs, 0.01H), 5.45 (brs, 0.01H), 4.58 (t, 0.02H), 4.07 (t, 1.89H), 2.33 (t, 2.00H), 1.67 (m, 4.26H), 1.41 (m, 2.21H) ppm.
PCL20-Ala2NH2 (4)
PCL20-Ala2Fmoc 3 (69 g) was dissolved in DCM (400 mL), and piperidine (5.0 g) was added. The reaction mixture was stirred at room temperature for 1 d and then poured into MeOH (1.5 L). The precipitate was re-dissolved in DCM, and the solution was again precipitated into MeOH. The precipitate was washed with MeOH and hexane and dried under high vacuum to yield PCL20-Ala2-NH2 4 as a colorless solid in a yield of 62 g (90%). 1H NMR (400 MHz, CDCl3): δ = 7.73 (brs, 0.01H), 4.59 (t, 0.01H), 4.09 (t, 2.06H), 2.34 (t, 2.00H), 1.67 (m, 3.98H), 1.41 (m, 2.34H) ppm.
mPCL
N-acetyl-l-alanine (1.76 g, 13.4 mmol), DIEA (2.16 g, 16.7 mmol), and PyBOP (7.56 g, 14.5 mmol) were added to a solution of PCL20-Ala2-NH2 4 (40 g) in THF (500 mL). The reaction mixture was stirred at room temperature for 5 d, then poured into MeOH (1.5 L). The precipitate was collected, washed with hexane, and dried under high vacuum to yield mPCL as a white solid in a yield of 39 g (98%). 1H NMR (400 MHz, CDCl3): δ = 6.60 (m, 0.02H), 6.13 (m, 0.01H), 4.47 (m, 0.04H), 4.09 (t, 1.90H), 2.33 (t, 2.00H), 2.04 (s, 0.07H), 1.67 (m, 3.94H), 1.41 (m, 2.14H) ppm; GPC: Mn = 22200 g/mol; Mw = 29300 g/mol; Ð = 1.32. TGA: decomposition onset: 387 °C. DSC: Tm = 54 °C, Tc = 28 °C, Td = 77 °C, Ta = 76 °C. The 1H NMR spectrum is shown in Supplementary Fig. S8.
Differential scanning calorimetry (DSC)
DSC measurements were performed with a Mettler Toledo DSC 3+ under N2 atmosphere (flow rate 10 mL min–1) at a scanning rate of 5 °C/min. Approximately 10 mg of mPCL was used for each measurement, which comprised four heating and three cooling scans to check for reproducibility. The results shown are from the second heating and first cooling scans.
Infrared (IR) spectroscopy
Films of mPCL were prepared for IR spectroscopy by drop casting from a 10 mg mL–1 solution in DCM onto KBr windows (iCL, REAL CRYSTAL IR CARD). The films were dried under vacuum, melted at 150 °C using a hot plate, and cooled to ambient temperature at 10 °C/min. The IR spectra were averaged over 100 scans recorded in transmission with a JASCO FT/IR 6300 spectrometer at a spectral resolution of 4 cm–1. The temperature was controlled with the PIKE accessory (Madison, WI) and measured by placing a thermocouple (METRA Instruments, Type-K) close to the specimen surface.
Oscillatory shear rheometry
Dynamic shear rheometry was performed with a TA Instruments ARES 2 rheometer and parallel steel plates. The diameters of the upper and lower plates were 8 and 25 mm, respectively, and the gap was set to 0.5 mm. To record temperature sweeps, molten mPCL was loaded at 150 °C and then cooled to 25 °C at 5 °C min–1, while applying a cyclic strain of amplitude 0.5% at a fixed angular frequency of 1 rad s–1. For the frequency sweeps, specimens were molten at 150 °C using a hot plate, and cooled to ambient temperature at 10 °C/min. The specimens were loaded at the experimental temperature, allowed to equilibrate for 15 min, and frequency sweeps were performed starting from 500 rad/s and scanning to 0.01 rad s–1. Time-temperature-superposition (TTS) was applied using the implemented software from TA Instruments to shift the curves horizontally by temperature-dependent shift factors aT, and vertically by bT.
Solution-phase NMR spectroscopy
Solution-phase 1D 1H NMR spectra were recorded in CDCl3 at 25 °C with a Bruker Avance III 400 spectrometer (9.7 T) at a 1H Larmor frequency of 400 MHz and calibrated with respect to the residual CDCl3 solvent peak (1H CDCl3 δ = 7.26 ppm).
Solid-state 1D NMR spectroscopy
For solid-state NMR spectroscopy, mPCL was melted in the oven of the ARES 2 rheometer at 150 °C and cooled to ambient temperature at 10 °C/min. Small fragments were cut from the solidified material and placed in a ZrO2 rotor with an outer diameter of 1.3 mm (2.5 μL specimen volume, filled to ~70%), which was subsequently sealed with SP1 bottom and drive caps from Bruker. Solid-state NMR experiments were performed with a Bruker Avance NEO 800 MHz spectrometer (18.8 T) equipped with a 1.3 mm standard bore CPMAS triple channel HCN probe. The temperature was controlled with the built-in control unit. After each change in temperature, the specimen was allowed to equilibrate for 5 min. The spectra were calibrated with respect to an adamantane reference specimen (1H adamantane δ = 1.82 ppm) at 25 °C, and the polymer h, i peaks at 60 °C (Fig. 2b). All spectra were recorded at a MAS frequency of 55 kHz. The longitudinal relaxation time (T1) was determined at 25 and 60 °C by fitting peak areas from saturation recovery experiments in order to determine the optimal recycle delay between scans (Supplementary Table S1). 1D 1H spectra were then recorded using a single-pulse sequence with a 90° pulse of 2.4 μs. The spectra were averaged over 16 and 512 scans at 25 and 60 °C, respectively, with the recycle delay set to ≥5T1. Pneumatic damping of the magnet was essential to reduce artefacts caused by external vibrations.
Solid-state CPMG NMR spectroscopy
CPMG experiments were carried out at a MAS frequency of 55 kHz using a CPMG49,50 pulse sequence with perfect echoes78, in which a 90° excitation pulse of length 2.4 μs was followed by a train of 180° echo pulses of length 4.8 μs and a 90° refocusing pulse of length 2.4 μs, with an echo delay time (τ), expressed as an integral number of rotor periods (rp) (Fig. 6). The recovery time was set to 1.3 T1. Pseudo 2D spectra were recorded for fixed τ and an increasing number (n) of echoes according to Table 1. The low concentration of the supramolecular end groups (2 wt%) meant that 128–512 scans were required for 1H detection at large τ and n. Four dummy scans were performed prior to each experiment, after which 512 transients were accumulated at 25 °C and 60 °C, respectively. The pulse sequences are available upon request.

CPMG pulse sequence with perfect echoes, in which an initial 90° excitation pulse is followed by a train of 180° pulses and a 90° refocusing pulse, separated by an echo delay time τ/4, resulting in a total echo delay time τ, and a total experimental time texp = τn.
Spectral processing and deconvolution
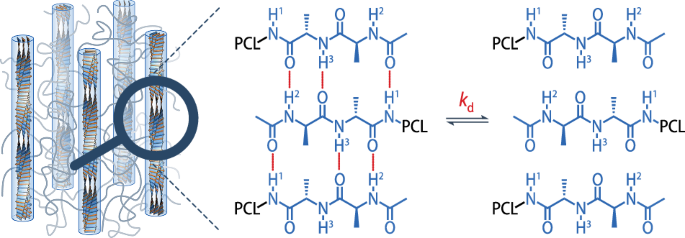
All spectra were processed with the TopSpin (Bruker) software, and the open source software DMFIT71 was used to deconvolute the processed spectra in the region δ = 6–12 ppm (end-group peaks) and δ = 0–6 ppm (polymer peaks). Table 2 gives the peak assignment and δ for all peaks used for the fits in the region δ = 6–12 ppm. For the spectra recorded at 25 °C, a total of seven peaks were selected for the deconvolution of the end group peaks: NH2 and NH3 between 8.7 and 10 ppm, NH1 at about 8 ppm, three NHweak peaks between 7.0 and 7.7 ppm, and the polymer peak c, which is assumed to account for overlap with contributions from the polymer backbone (Fig. 3b). The assignment of NH1, NH2, and NH3 was based on solution-phase spectra, in which the amides gave rise to two overlapping peaks (NH2 and NH3) and a third, well-separated peak (NH1) shifted by about δ = –0.5 ppm relative to the former peaks (Fig. 2b). The NHweak peaks were identified from solid-state spectra recorded at various temperatures between 25 and 60 °C, which showed the broad peak present at δ = 7.0–7.7 ppm at 25 °C (Supplementary Fig. S7) to transform into three distinct, sharp peaks in the same region at temperatures above about 50 °C. In the spectra recorded at 60 °C, the polymer peak c no longer extended beyond δ = 6 ppm, because the PCL matrix was molten and its FWHM was consequently reduced. Moreover, there was no clear evidence of a broad NH1 peak in these spectra, presumably because dissociation occurred at a timescale of τ < 4 rp. We therefore reduced the number of peaks used for the deconvolution of the spectra recorded at 60 °C to five: NH2 and NH3 at 9.6 ppm and 9.0 ppm, and the three NHweak peaks at δ = 7.3–7.6 ppm.
A Gaussian fit was used for the NH2 and NH3 peaks, and a Lorentzian fit for all other end-group peaks. The peak positions were assumed to remain unchanged after the first fit for each temperature and τ dataset, while the FWHM and amplitude were adjusted. To deconvolute the polymer backbone peaks c, e, h, i, and j in the spectral region δ = 0–6 ppm, a combined Gaussian and Lorentzian fit was used for each peak at 25 °C, and a single Lorentzian fit for peak d, resulting in a total of nine peaks (h and i appear at the same position). At 60 °C, Lorentzian fits were sufficient for peaks c, d, e, h, i, and j, because the PCL matrix was fully molten.
Fitting to the Carver-Richards Model
To obtain dissociation rate constants, kd, we fitted the R2 data obtained from the CPMG experiments for the peaks assigned to NH2 + NH3, to the Carver–Richards equation73,74,75. In the case of a fast two-site exchange between aggregated (A) and dissociated (D) state of the amide moieties, the Carver–Richards model simplifies to Eq. 2, where R2,0 is the transverse relaxation rate in the absence of dynamics, kd is the rate constant for the exchange from state A to D, and ɸ = pA∙pD∙Δω2, where pA and pD are the fractions of aggregated and dissociated amide groups, respectively, and Δω is the difference in resonance frequency of the peaks associated with the two species73. In absence of well-distinguishable peaks for the two states, indicating a fast exchange according to Carver–Richards, these latter three parameters cannot be obtained separately and are thus combined in ɸ.
2D exchange spectroscopy experiments
The exchange spectroscopy (EXSY) experiment was conducted on a 900 MHz Bruker wide bore spectrometer (21.1 T) equipped with an Avance Neo console and a 1.3 mm HX double resonance CPMAS probe. The samples were packed into 1.3 mm zirconia rotors under ambient conditions. The rotor was spun at 55 kHz spinning speed using dry nitrogen gas. The pulse sequence consisted of three 90° pulses of 1.0 μs which were separated by an incremented evolution period (rotor synchronized increments of 67 μs) and a mixing time of 20 ms. 128 transients were cumulated for each of the 910 slices constituting the indirect dimension. The recycle delay was set to 3.6 s, which is equivalent 1.3T1 of the slowest relaxing species.
link