Materials
The polysaccharide was purchased from the local supermarket.
Preparation of acetylated Wendan polysaccharide
0.5 g of PP-2 was weighed and dissolved in 10 mL of distilled water. The polysaccharide solution was transferred to a round bottom flask, and 0.5 mol/L NaOH was gradually added to adjust the solution until the pH was 9.0. During this period, a magnetic stirrer was used to continuously stir. At 30 °C, the mixture was slowly added with 0.6 mL of acetic anhydride and NaOH solution alternately, while maintaining the pH of the solution at 8.0–10.0. After the solution is added, seal and react at a constant temperature for 2 h. After the reaction is complete, adjust the pH with dilute HCl solution to make the solution neutral. After the reaction solution cools down, add anhydrous ethanol while stirring, and let it stand at 5 °C in the refrigerator for 24 h. After precipitation in the solution, centrifuge the mixture at 5000 r/min for 10 min to retain the precipitate. Dissolve the obtained precipitate in ultrapure water, then place it in a dialysis bag and seal it with a sealing clip. Dialysis with flowing water for 48 h, followed by dialysis with distilled water for 24 h. After lyophilization, acetylated Wendan polysaccharides can be obtained.
Determination of substitution degree of acetylated Wendan polysaccharide
Titration method is used to determine the degree of acetyl substitution, with slight modifications based on reported literature7. The specific steps are as follows: Dissolve 10 mg of Ac-PP in a three necked flask, add 10 mL of NaOH with a concentration of 0.01 mol/L, and sonicate for 5 min to fully dissolve it. After the reactants are fully dissolved, fix the three necked flask in a 50 °C constant temperature water bath and vigorously stir for 2 h using a magnetic stirrer to ensure complete saponification. After the reaction is complete, cool the solution to room temperature and add phenolphthalein indicator dropwise. The solution appears pink. Finally, the solution is titratedwith 0.01 mol/L HCl until the pink color disappears. The degree of substitution is calculated according to the following formula:
$$\text{DS} = \frac{132A}{4300 – 42A}$$
(1)
In Eq. (1), DS represents the degree of acetyl substitution; A represents acetyl content:%.
Preparation of phosphorylated Wendan polysaccharides
Seven grams of sodium tripolyphosphate and sodium trimetaphosphate (in a ratio of 6:1) were weighed. Add 100 mL of distilled water and sonicate to dissolve in a three necked flask. Subsequently, 0.5 g of PP and 5% sodium sulfate (10 mL) were added to the three necked flask. After the reactants were fully dissolved, the pH of the solution was adjusted to 8.5. Place the three necked flask in a water bath for phosphorylation reaction. The temperature is set to 80 °C and the reaction time is 4 h. After the reaction is completed, the reaction solution is concentrated by rotary evaporator and then placed in a dialysis bag for 2 days of dialysis with flowing water. Finally, the dialyzed solution is collected and freeze-dried to obtain P-PP.
Determination of phosphate substitution degree
The determination of phosphate content was carried out using the ammonium phosphomolybdate method8, and then the degree of substitution was calculated based on the phosphate content.
$$\text{DS} = \frac{5.23\text{P}}{100 – 3.32\text{P}} $$
(2)
In Eq. (2), P represents the percentage (%) of phosphate groups in P-PP.
Preparation of carboxymethylated Wendan polysaccharides
0.5 g of Wendan peel polysaccharide was dissolved in 40 mL of 20% NaOH solution, and then stirred in a magnetic stirrer for 1 h. Then slowly add 60 mL of isopropanol and 2.4 g of chloroacetic acid, and continue stirring the reaction at 55 °C for 4 h. After the reaction is complete, let the reaction mixture stand and cool. Adjust the pH of the reaction solution with dilute hydrochloric acid until neutral. Subsequently, collect the reaction solution and dialyze it with flowing water for 2 days and deionized water for 1 day. After dialysis, add ethanol (3 volume) to the reaction solution (1 volume) to produce a precipitate. Collect the sediment and freeze dry it to obtain carboxymethyl Wendan peel polysaccharide.
Determination of degree of substitution of carboxymethyl Wendan polysaccharides
The degree of substitution of carboxymethyl Wendan peel polysaccharides was determined using acid-base titration method9. Accurately weigh 30 mg of CM-PP, transfer it to a beaker, and add 75 mL of sodium hydroxide solution (0.1 mol/L). Put the mixed solution into a magnetic stirrer and stir until the polysaccharides are completely dissolved. Add phenolphthalein dropwise to the solution and titrate the solution back using 0.1 mol/L HCl as the titrant. The calculation of the degree of substitution of carboxymethyl Wendan peel polysaccharides is carried out according to the following formula:
$$\text{DS} = \left( \text{M}_{\text{NaOH}} \times \text{V}_{\text{NaOH}} – \text{M}_{\text{HCl}} \times \text{V}_{\text{HCl}} \right) \times \frac{162}{\text{W}_{\text{polysaccharide}}} $$
(3)
M is the molar concentration, V is the volume, W is the mass.
Determination of polysaccharide content
The content of polysaccharides was determined using the phenol-sulfuric acid method10. 1mL of 5% phenol solution and 5mL of concentrated sulfuric acid were quickly added to the polysaccharide solution, thoroughly mixed and shaken. Subsequently, the mixed solution was reacted in a 60 °C water bath for 15 min. After cooling the mixture to room temperature, the absorbance at 490 nm was measured using a UV visible spectrophotometer (UV-2550, Shimadzu). Among them, distilled water was used as the blank control group for the experiment. Finally, the glucose standard curve was plotted to calculate the total sugar content of polysaccharides, with the x-axis representing glucose concentration and the y-axis representing absorbance.
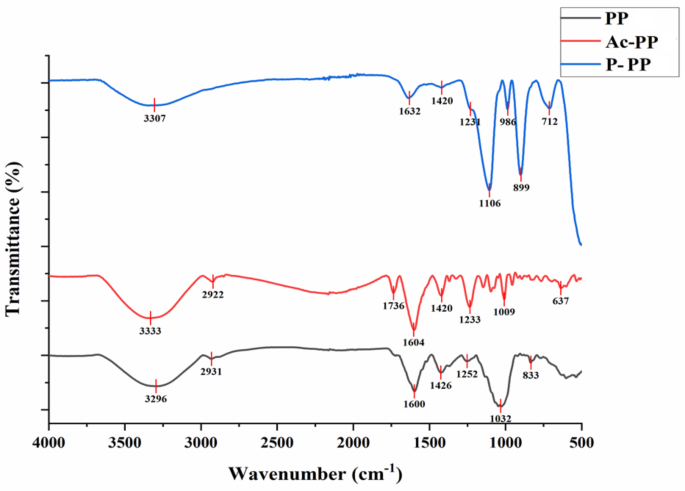
Infrared spectroscopy measurement
After drying, KBr was mixed with an appropriate amount of polysaccharide sample in an agate mortar and ground evenly. Then the mixture was compressed into flakes, and the sample could not be loose. The sliced sample was placed in an infrared spectrometer (Spectrum Two, PerkinElmer) for testing and scanned in the range of 4000 –500 cm− 1 to obtain an infrared spectrum.
NMR measurement
Approximately 50 mg of polysaccharide sample was weighed and dissolved in heavy water (600 µ L) as a solvent. The solution was carefully aspirated and transferred to a nuclear magnetic resonance tube. The nuclear magnetic resonance spectrum was obtained using a 300 MHz nuclear magnetic resonance spectrometer (AVANCEII, Bruker). The 13C NMR and 31P NMR spectra were recorded, respectively.
Determination of hydroxyl radical scavenging ability
The hydroxyl radical scavenging ability is an important indicator for measuring the antioxidant capacity of substances, therefore measuring the hydroxyl radical scavenging ability of PP and its derivatives is of great significance. The hydroxyl radical scavenging ability is based on the method proposed by He et al. and has been modified accordingly11. Firstly, 1mL of PP, Ac PP, P-PP solutions, and CM-PP aqueous solutions with different concentrations (0 mg/mL, 0.1 mg/mL, 0.2 mg/mL, 0.4 mg/mL, 0.8 mg/mL, 1.6 mg/mL, 3.2 mg/mL) were prepared, and 1 mL of pre prepared FeSO4 (8 mmol/L), salicylic acid ethanol solution (8 mmol/L), and H2O2 (8 mmol/L) were added sequentially. The mixture was placed in a constant temperature water bath and reacted at 37 °C for 30 min. The absorbance As at 510 nm was measured. Secondly, deionized water was used instead of H2O2 to measure the background absorbance Ar of the corresponding test solution. Deionized water was used instead of the test solution, and the absorbance A0 of the control group solution was measured. Using Vc as the positive control, each experiment was measured in parallel three times. The formula for calculating clearance rate was as follows:
$${\text{Hydroxyl\;radical\;scavenging\;rate }} (\%) = [1 – (A_s – A_r)/A_0] \times 100$$
(4)
Determination of DPPH radical scavenging ability
A DPPH anhydrous ethanol solution with a concentration of 0.1 mg/mL was prepared. The polysaccharide aqueous solution was diluted to different concentrations (0 mg/mL, 0.1 mg/mL, 0.2 mg/mL, 0.4 mg/mL, 0.8 mg/mL, 1.6 mg/mL, 3.2 mg/mL), and 1 mL was taken and mixed with the DPPH-anhydrous ethanol solution. The mixture was reacted under completely dark conditions for 40 min, and then a sample was taken to measure the absorbance (A0) value at 517 nm. The experiment needed to be repeated three times with Vc solution as the positive control group. The DPPH radical scavenging activity was calculated according to the following formula:
$$\text{DPPH\;scavenging\;activity } (\%) = [1 – (A_0 – A_1) / A_2] \times 100$$
(5)
In the above equation, A0 represents the absorbance of the sample; A1 represents the absorbance of PP solution and anhydrous ethanol; A2 represents the absorbance of deionized water and DPPH.
Determination of superoxide anion radical scavenging ability
Firstly, 1mL of polysaccharide aqueous solution (0 mg/mL, 0.1 mg/mL, 0.2 mg/mL, 0.4 mg/mL, 0.8 mg/mL, 1.6 mg/mL, 3.2 mg/mL) was precisely transferred into a test tube, and then 3 mL of Tris-HCl solution (pH = 8.2) was added. The mixed solution was placed in a water bath and immersed in water at 25 °C for 15 min Subsequently, 12 µ L of pyrogallic acid (3 × 10− 3mol/L) and 0.5 mL of concentrated HCl were added to the mixed solution. Finally, the absorbance of the mixed solution was measured at a wavelength of 325 nm. The superoxide anion scavenging capacity was calculated as follows:
$$ {\text{Superoxide }}\;{\text{anion }}\;{\text{scavenging}}\;{\text{ capacity}}\;{\text{ }}\left( \% \right){\text{ }} = \left[ {\left( {{\text{A}}_{{\text{1}}} – {\text{A}}_{{\text{2}}} } \right)/\left( {{\text{A}}_{{\text{1}}} – {\text{A}}_{{\text{3}}} } \right)} \right]{\text{ }} \times 100 $$
(6)
In the formula, A1 was distilled water as the blank group; A2 was the experimental group; A3 with buffer solution as the control group corresponding to the absorbance value.
Statistical analysis
All experiments were conducted in parallel three times, and the experimental data were analyzed using SPSS 28 (IBM). p < 0.05 was considered statistically significant.
link