Soda lignin as a sustainable photosensitive component for conventional and controlled radical photopolymerization
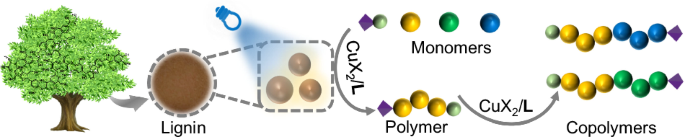
Motivation and analysis of AL
This study focused on different protocols following either conventional radical polymerization or radical photopolymerization with ≲50 ppm CuBr2/L according to a photo-ATRP setup40,46,47,48 with a 420 nm emitting LED. The increased reports of toxicological issues of traditional radical photoinitiators38,39 have initialized the development of alternative materials49 where fewer problems would be expected. Lignin appears as a reasonable substrate since its biological origin classifies it as a material with no toxicological issues. Motivated by this, attention has been paid to the valorization of technical lignin available as waste; that is soda lignin/alkaline lignin (AL).
AL mostly operated as heterogeneous material dispersed in the reaction mixture, Fig. 1a. The size was between 100-300 nm. Figure 1b illustrates a general pattern of lignin with some respective structures. The complex supramolecular structural features formed by the aromatic moieties can be seen as one source to initiate photonic events; that is for example, long afterglow18,19,20,22,50. Thus, the potential of lignin to sensitize radical photopolymerization can be seen as a logical consequence16,18,19,51. It can operate in a photocatalytic cycle following a protocol based on light-mediated Atom Transfer Radical Polymerization (photo-ATRP)40,46,48, where photoinduced reduction of the deactivator (CuX2/L) mainly controls the efficiency of activator (CuX/L) formation. Figure 1c exhibits the necessary chemicals for this protocol comprising ethyl α-bromophenylacetate (EBPA), the cocatalyst CuBr2/L operating as deactivator complexed with the amine ligand L (L: Me6TREN, PMDETA: N, N, N′, N′′, N′′- Pentamethyl diethylenetriamine) to make it compatible in the respective monomer/solvent mixture. The structural features shown in Fig. 1d additionally facilitated aryl sulfonation, resulting in a one-component photoinitiating system following Norrish I cleavage. The low ability to initiate conventional radical required this modification.
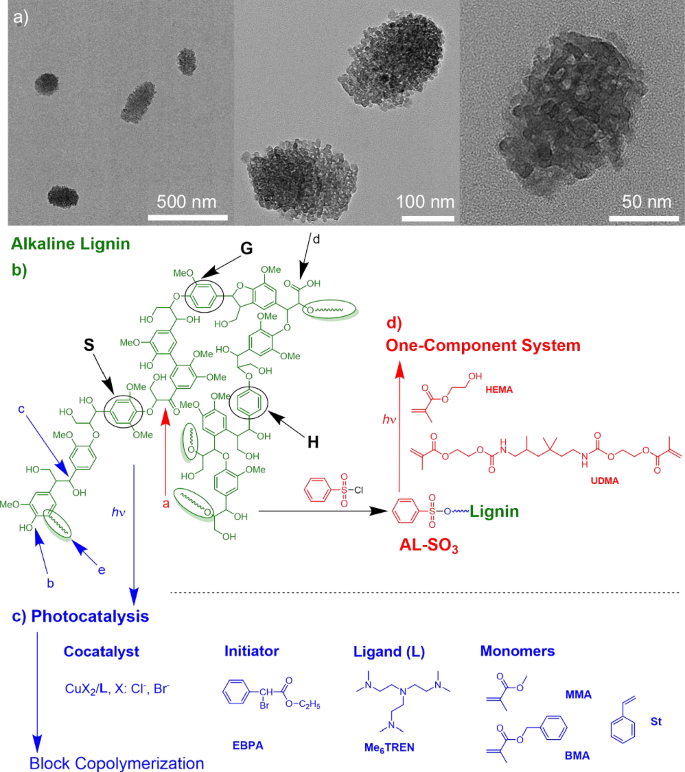
a Transmission electron microscopy (TEM) of AL in dimethyl sulfoxide (DMSO) with different magnifications. b Sketch for alkaline lignin. c Photoexcited AL additionally enables radical polymerization based on a photocatalytic mechanism with alkyl bromides as initiator and CuBr2/L as cocatalyst. Me6TREN was the ligand L. d Functionalized AL (AL-SO3) initiates conventional radical photopolymerization of urethane dimethacrylate (UDMA) and hydroxyethyl methacrylate (HEMA) as one-component photoinitiator (AL-SO3).
X-ray photoelectron spectroscopy (XPS) survey (Fig. 2a) showed C and O as major elements. Sodium originated from the paper-making process. High-resolution XPS exhibited four peaks in the C1s spectrum (Fig. 2b) appearing at 284.8 eV, 286.3 eV, 288.3 eV and 289.1 eV assigned to C-C, C-O-C, C=O and O-C=O moieties, respectively. Signals at 530.9 eV, 531.7 eV, and 535.4 eV in the O1s spectrum (Fig. 2c) related to C=O (carbonyl), C-OH/C-O (hydroxyl, ether), and O-C=O (ester) moieties, respectively, in AL. The FTIR spectrum (Fig. 2d) showed a broad band indicating the OH vibration (~3359 cm−1). The vibration at 1120 cm−1 is related to the C-O-C ether structures and characteristic peaks (~1600 and ~1510) of the benzene ring.
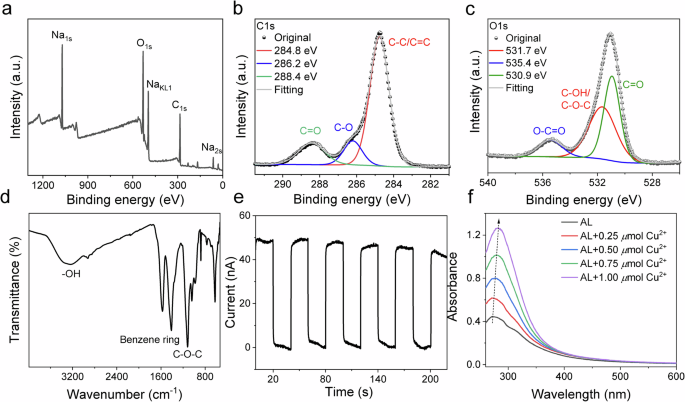
a XPS survey spectrum; b, c high-resolution XPS spectra of C1s and O1s; d FT-IR spectrum of AL; e photocurrent of AL under xenon lamp; f UV-Vis absorbance spectra of AL and loading of different content of CuBr2.
In addition, photocurrent experiments (Fig. 2e) did not give any evidence regarding the formation of long-living charges in the dark period52,53,54. Room temperature phosphorescence (RTP), and therefore the formation of triplet states, was not observed either. Thus, the paper manufacturing process can be seen as a one reason to contribute for the loss of supramolecular structures due to the alkaline treatment. Supramolecular structures contribute to long afterglow and thus to the formation of triplet states18,19,20,22,50, and charge migration events52,53. Absorption spectroscopy demonstrated an increase of absorption upon the addition of CuBr2 (Fig. 2f) indicating binding/interacting with lignin.
Absorption spectroscopy and electrochemical properties answered questions related to the absorption efficiency and capability to donate an electron in photoredox cycles (Figs. S1a, b for more details in Supplementary Information). Cyclic voltammetry (CV) analysis showed an oxidation peak (Eox) at 1.15 V in the case of AL (4 mg/mL), which gradually shifted to lower values after the addition of Cu(II); that is an Eox of 1.09 V with 25 ppm CuBr2/Me6TREN, 1.05 V with 52 ppm CuBr2/Me6TREN), and 0.99 V with 100 ppm CuBr2/Me6TREN), see Fig. S1c in Supplementary Information. This shows an increase of the oxidation capability of lignin moieties interacting with CuBr2. Switching to CuCl2/Me6TREN connected to an additional decrease of Eox of 0.87 V with 31 ppm loading of CuCl2/Me6TREN. Reduction showed a change of the Ered peak to −0.7 V after adding of 31 ppm CuCl2/Me6TREN to AL (Figs. S2–3 in Supplementary Information). These data are needed for the calculation of the free reaction enthalpy of the photoinduced electron transfer (ΔGel) vide infra according to Eq. 1 (F = Faraday constant, E00 = excitation energy).
$$\Delta {{{\rm{G}}}}_{{{\rm{el}}}}=F\times ({E}_{{{\rm{ox}}}}{-}{E}_{{{\rm{red}}}}){-}{{{\rm{E}}}}_{00}$$
(1)
The ΔGel calculation resulted in a negative value for the reaction of the photoexcited donor AL and CuBr2/Me6TREN operating as an oxidizing agent. AL, depending on the loading with CuBr2/Me6TREN described vide supra an oxidation potential with an oxidation potential between 1.15 V to 0.99 V, an E00 value of 2.36 eV (525 nm), and a reduction potential of –0.39 V55 for (Cu/Me6TREN)Br2. The ΔGel resulted between −0.82 eV to −0.98 eV, respectively. This quantity indicated that photoinduced electron transfer (PET) can proceed from a thermodynamic point of view, where lignin operates as a photosensitizer to initiate the photocatalytic cycle of the ATRP. This does not include considerations of internal activation barriers56 in the PET protocol. The redox potentials depend on the concentration of CuBr2/L affecting somehow the standard potential. This also affects the ATRP equilibrium constant discussed vide infra. A lower copper concentration may drive the scenario towards chain termination and therefore increase of dispersity. Thus, one needs to compromise between the required uniformity of the polymer and the acceptable amount of copper catalyst.
Feasibility to operate as a one-component photoinitiator
Previous studies of lignin reported about the availability of Aryl-CO-R moieties in lignin (Scheme 1)57. This group can homolytic cleave, resulting on Aryl-CO∙ and R∙, which shall initiate conventional radical polymerization. It should enable this biomaterial as a source to generate initiating radicals following Norrish I cleavage resulting on enabling this material as a biobased one-component initiator. However, the results obtained evidenced no effective initiation of conventional radical photopolymerization using a monomer mixture consisting of HEMA and UDMA. Here, the addition of HEMA improved the performance to disperse lignin in UDMA. Nevertheless, AL exhibited insufficient initiation efficiency in the HEMA/UDMA mixture. However, these radicals have a lower mobility because they originate from polymer matter and should therefore result in a significantly lower initiation efficiency. The higher molecular weight of these radical connects to lower diffusion constants and there less reactivity. More target-oriented was the reaction between AL and benzene sulfonyl chloride, resulting in aryl sulfonic esters (AL–SO2Ph). Here, bond cleavage can successfully form low molecular PhSO2∙ radicals exhibiting higher mobilty49. This could be a feasible approach to compensate the drawbacks vide supra. Such a treatment additionally increased the hydrophobicity, which connects to an improved dispersibility of AL–SO2Ph in the crosslinking monomer mixture. Its exposure resulted in conventional radical photopolymerization of HEMA/UDMA, Fig. 3. A decrease in initiator loading did not strongly affect the final version, which appears more typical for traditional photoinitiators43. Such systems are much more complicated than low molecular weight photoinitiators because the heterogeneous nature of such heterogeneous systems can lead to composite-like materials after irradiation. This has been shown in a previous study. A forthcoming study will report on the progress in this field.
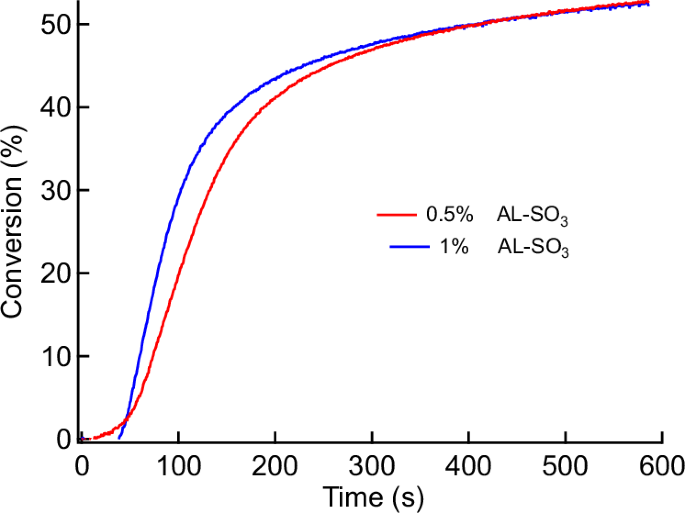
Double bond conversion of aryl sulfonated lignin (AL–SO3) in radical polymerization using a mixture of HEMA and UDMA (λ = 365 nm, I = 35 mW/cm2).
AL without functionalization demonstrated no notable activity under our experimental conditions, despite its photocleavable groups. This is likely lower compared to those photoactive groups obtained after functionalization. Functionalization of the phenolic OH groups with photocleavable moieties is clearly beneficial because it reduces the number of hydroxyl groups in the photoactive material, thereby minimizing interference from conventional radical polymerization. Again, this preliminary result demonstrates the potential of AL to serve as natural feedstock to design photoinitiating one-component photo-initiating materials but it keeps in mind that more research will be required to discover the full potential.
Operation as photosensitizer in photocatalytic cycles for photoinitiation
A photo-ATRP protocol, as shown in Fig. 4a, served as the fundamental to design the photocatalytic system (Eq. 2a–d)58. According to the spectroscopic results disclosed in Figs. S4–7 in Supplementary Information, additional interactions shall be included that relate to the adsorption/desorption of CuBr2/L on the surface of AL (Eq. 3). This process needs more consideration in future works because it also relates to reactivity and reaction conditions of the ATRP proceeding nearby the particle. Such explorations can be still seen its infancy although some pioneering works already appeared. In addition, the supramolecular structure relating to photoreactivity19,22 may depend on the lignin batch, which related to waste in this study40,41,42,52,53,59. Thus, the source and method to get it may additional parameter determining microstructure of the surface.
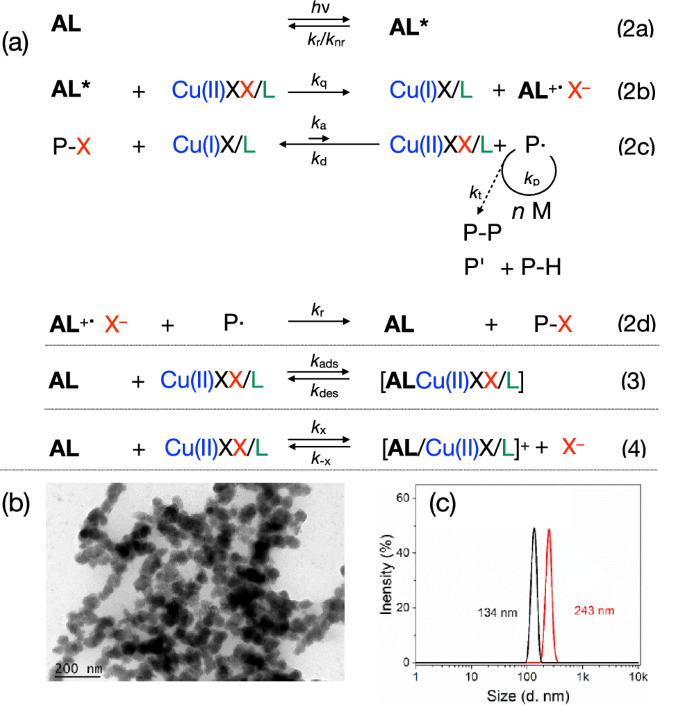
a Sensitized light-mediated ATRP with traces of deactivator (CuBr2/L) in the ppm region (ka = activation rate constant, kd = deactivation rate constant, kp = propagation rate constant), modified from ref. 58. This equation importantly affects the efficiency of photo-ATRP also in heterogeneous systems. Here, additional conditions must be considered that are influenced by different interactions with lignin. This includes: (1) full exchange between Br– and phenolate, (2) partial exchange between Br– and phenolate, (3) no exchange between Br– and phenolate. (4) relates to metal-free photo-ATRP based on an oxidative mechanism. b TEM picture of an isolated AL particle after photo-ATRP with MMA. c particle size of AL before (black) and after (red) photo-ATRP with MMA.
Ion exchange by phenolic groups (Eq. 4) may be one possibility influencing the photolytic rate of activator formation and deactivator back formation (Eq. 2b) followed by chain propagation (Eq. 2c). Binding of reactants on the surface would also impact the reactivity because adsorption effects mobility, and ion exchange the equilibrium constant of ATRP (KATRP). Figure 4b shows a TEM picture of an AL particle isolated after photopolymerization. It exhibits a larger size after the reaction, as approved by light scattering measurements, see Fig. 4c. The inclusion of a swollen polymer into the free spaces of the particle may additionally explain this phenomenon. This clearly increases viscosity around the particle surface because there is less efficient mixing with the solvent. Different reactivity conditions exist because the reactions proceeding in the cavities of the particle are different from the reactions in the surroundings outside of the particle.
Table 1 summarizes the polymerization results vide infra. Entries 1-3 show a decrease in conversion, the number average molecular weight (Mn), and initiation efficiency Ieff60 with increasing initiator concentration, while the dispersity (Đ) only slightly responded. Repeating experiments confirmed reasonable reproducibility. Furthermore, a higher AL concentration (Run 4) resulted in higher conversion while the dispersity increased, confirming less uniformity. A lower AL loading, as shown in run 5, resulted in less conversion and lower Ieff. The changed absorption conditions mainly affected these changes compared with run 1. Dispersity increased with decreasing L and CuBr2 to 25 ppm (run 6), while increasing L and CuBr2 to 102 ppm (run 7), Mn remained like run 6 but the conversion was the largest.
A higher concentration on initiator and deactivator, namely CuBr2/Me6TREN, in run 8 resulted in lower conversion, Mn, and Đ. A change of MMA by Styrene (St) led to a lower conversion while the Đ exhibited the best uniformity of molecular weight distribution (run 9). Change of L by PMDETA (run 10) approved less uniformity and Ieff compared to run 1. The reduction potential of EBPA should enable PET with photoexcited AL, resulting in initiating radicals that shall deactivate with less selectivity in an oxidative metal-free photo ATRP scheme53,61. Surprisingly, additional experiments without copper catalyst and L (run 11 and 12) resulted in polymers accompanied by high conversion and large Đ with no possibility of extending the chain, see Fig. S8 in Supplementary Information. Thus, chain termination efficiently competed and assigned both examples to conventional radical photopolymerization. Here, the alkyl bromide operated as a coinitiator following a classical multi-component photoinitiator setup. Nevertheless, such a system may appear interesting for several uses in practice due to the low toxicity of AL and the macroinitiator because both are polymers with low migration potential. In addition, a comparative experiment (run 13) without AL showed no polymerization, confirming that the excitation of CuBr2/Me6TREN does not have an impact on the results here. Apparently, the addition of (Cu/Me6TREN)Br2 suppresses the dominance of chain terminations as documented by the lower dispersity. On the other hand, photopolymerization with AL and FeBr3 resulted in a polymer with much higher Mn and Đ, indicating the dominance of chain termination and therefore conventional radical polymerization. Thus, choosing (Cu/Me6TREN)Br2 as a cocatalyst was the more effective option to suppress chain termination, although other systems showed more promising results62. It could be considered as an alternative, although compromises need to be made regarding the control of the polymerization process. The use of Fe-based cocatalysts would allow a wider application in technologies, as Cu-based cocatalysts require processing in reactors with less tendency.
Runs 1, 4, 5, and 6 were considered to check out the lowest amount of deactivator needed to operate in the photo-ATRP setup before it switches to conventional radical photopolymerization, caused by the domination of dead chain formation. Runs 14-17, which correspond to a Cu(II) loading between 52-6.5 ppm, show an increase in dispersity with decreasing deactivator concentration. Nevertheless, all polymers obtained successfully showed chain extension, although the amount of non-extendable chains increases with decreasing deactivator loading vide infra.
In general, such materials can be considered as a composition of polymers comprising chain extendable polymer chains comprising bromine and those with dead chains formed by termination. This depends on the loading with the deactivator. For example, an additional increase of AL slightly decreased Đ, which exhibited the lowest value in run 18 of all experiments shown in Table 1. An additional increase of AL (runs 19 and 20) did not significantly impact polymer data compared to run 18. Isolation of AL after polymerization indicated nearly no bound Cu(II) on the surface of AL while complementary experiments with FeBr3 did (Fig. S9 in Supplementary Information). Complexation with Me6TREN results in a much better complex stabilization, explaining the lower deposition in the AL surface.
These findings additionally motivated to check whether CuCl2 also operated in this setup. Runs 21-24, representing loadings of CuCl2 between 31 and 2 ppm, approved this showing a clear increase of Đ and, therefore, also decreasing uniformity with decreasing CuCl2 loading. This appears to be more significant with CuBr2 as additionally shown by the block copolymerization experiments vide infra. A 2 ppm loading of CuCl2 (Run 24) resulted in PMMA with a yield of 49%, Mn of 54.2 kDa, and dispersity of 2.45. However, this material also contains chain extendable polymers as demonstrated by the increase of Mn by 7 kDa with no significant change of the dispersity vide infra. Thus, the number of polymers contributing to chain extension can be seen as small.
As expected, changing the monomer to Styrene (run 25) resulted in less conversion due to monomer’s less reactivity. Nevertheless, the polymer obtained was transferred to block copolymerization experiments either, where it operated as a macroinitiator vide infra.
A kinetic study confirmed a linear increase in conversion during exposure (Fig. 5a). Temporal control of these experiments was performed by switching the blue light LED ON/OFF intermittently (Fig. 5b). The system showed no response in the dark, indicating the back-formation of the deactivator CuBr2/Me6TREN in the catalytic exposure cycles.
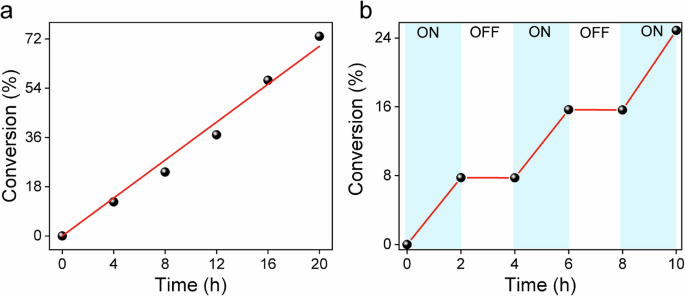
Reaction condition: [MMA]/[EBPA]/[CuBr2]/[Me6TREN]=300/2/0.03/0.135 in DMSO 75 vol% (mAL = 4.5 mg). a Kinetics of the photopolymerization, b Temporal control of the photoinduced ATRP process.
Chain end fidelity
To verify the end-group fidelity of polymers, chain extension experiments were carried out with further monomers. The macroinitiator PMMA-Br successfully extended its chain with MMA, benzyl methacrylate (BMA), and Styrene (St). Complementary experiments with different deactivator loading also approved the feasibility of chain extension, while the fraction of polymers with dead chains increased with decreasing metal halide concentration, as shown by the increase of Đ. Figure 6 illustrates the scenario using either CuBr2 or CuCl2 as a co-catalyst. In the case of CuBr2, the second block exhibited a higher dispersity, Fig. 6a, although both blocks possessed a high average polymerization degree Pn. The change of the second block to BMA, Fig. 6d, gave a similar scenario considering the Pn of both blocks. The use of St resulted in the expected results, where the block of poly-St exhibited a lower polymerization degree in both examples, Fig. 6e–f. The use of poly-St as a macroinitiator, Fig. 6f, clearly demonstrates its high reactivity as shown by the larger Pn of the second block; that is b-PMMA. This agrees with previous findings using heterogeneous photocatalysts in photo-ATRP cycles41,42,52,53. However, a lower loading with copper also led to a higher polymerization degree caused by the competing contribution of conventional radical polymerization.
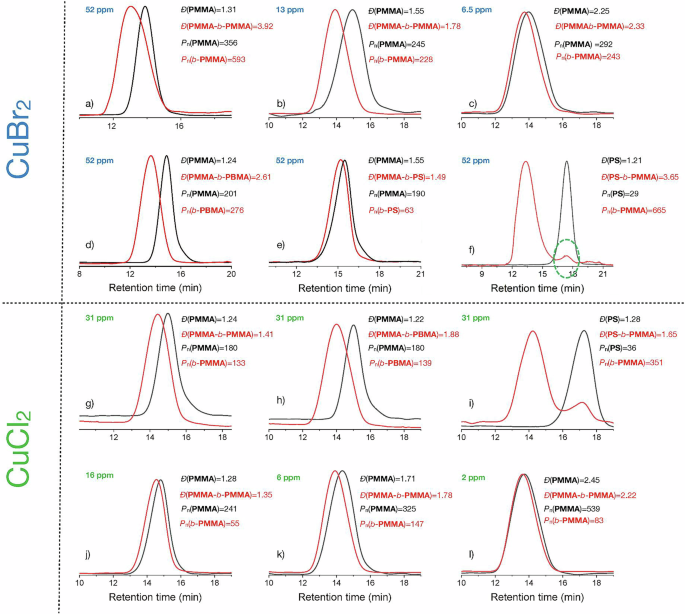
Reaction condition: [MMA]/[EBPA]/[CuX2]/[Me6TREN]=300/2/0.03/0.135 in DMSO 75 vol% (mAL = 4.5 mg). GPC of PMMA and chain-extended PMMA (second block: b-PMMA) with different loading of CuBr2 deactivator, a 52 ppm; b 13 ppm; c 6.5 ppm. Chain extension with different monomers (52 ppm CuBr2). d PMMA with BMA (second block: b-BMA); e PMMA with St (second block: b-PS); f PS with MMA (second block: b-PMMA). Using CuCl2 as deactivator (31 ppm loading) and different monomers results in the GPC shown in g PMMA and chain-extended PMMA (second block: b-PMMA); h PMMA and chain-extended PMMA with BMA (second block: b-BMA); i PS and chain-extended PS with MMA (second block: b-PMMA). GPC of PMMA and chain-extended PMMA (second block: b-PMMA) with different loading of CuCl2 deactivator. j 16 ppm; k 6 ppm; l 2 ppm.
Further experiments should focus on getting a more detailed pattern of the end group. This responsibility operates in the chain extension experiment. These can be mass spectrometric experiments, such as matrix-assisted laser desorption ionization (MALDI) mass spectrometry, which are particularly useful for halogen atoms as a narrow group, where the isotopes give a specific pattern to the molecular ion. A recent report of photo-ATRP focused on these aspects63,64. This gives a detailed pattern about the availability and chain-extendable groups comprising bromine and those ending in dead chains. The interpretation of MALDI mass spectrometric data also requires a critical interpretation of the peak intensities.
Furthermore, a detailed analysis of the NMR spectra with respect to the end group was not pursued because the degree of polymerization was too high to obtain a reliable number related to Mn. Figure S10 in Supplementary Information shows the NMR spectra for the two copolymers of Fig. 6d and 6e with respect to PMMA-b-PBMA and PMMA-b-PS, respectively. Based on the data obtained from the experiment, no further evaluation was carried out to avoid over-interpretation of the results. This worked better in a previous report64. The molecular weights reported there remained on the lower region, which enabled a more reliable data evaluation because the respective end group peaks exhibited a much larger intensity based on the signal/noise ratio.
photo-ATRP under air
Experiments were carried out under air. The concentrations of the ATRP cocktail were taken from Run 14 in Table 1 and sodium pyruvate (Py), CH3-COO– Na+, was added to deactivate inhibiting oxygen in the system (required filtering of precipitated NaBr of the reaction cocktail prior to use) following a previous procedure of a non-sensitized CuBr2/L-based photo-ATRP65,66. Tetrabutylammonium bromide (TBAB) addition increased bromide concentration65,66. Alternatively, addition of 15-crown-5 (15-5) should complex Na+ to prevent filtration. Table 2 shows the results obtained.
The polymer obtained exhibited a dispersity of 1.32 (Run A). Thus, ion exchange in CuBr2/L by Py, disclosed earlier65 followed by filtering was effective. Complexation of Na+ by 15-5 did not bring the expected reactivity (Run B). Run C demonstrated failure after adding of TBAB. Experiments pursued as sealed samples with no inertization exhibited similar findings as documented by Run D. Run E resulted in a polymer with much higher dispersity and conversion. A higher contribution of conventional radical polymerization explains these findings. Obviously, 15-5 interfered with the ATRP cycle, Run F.
A mechanistic discussion should be based on the conversion of inhibiting oxygen into those with less inhibiting properties, such as O2–• following a previous proposal to pursue oxygen-tolerant metal-free ATRP67. As shown vide supra, the metal-free ATRP did not work with AL. It succeeded better even in photo-RAFT systems29,68. Thus, CuBr2/Me6TREN was added as cocatalyst. Processing under air requires extension of this proposal. The addition of Py enabled the conversion of inhibiting oxygen into an oxygen species with less inhibiting properties in the reaction cocktail. This is related to a Fenton cycle69 where the activator formed and oxygen resulted in the formation of deactivator, and the less-inhibiting superoxide anion O2-• and H2O265,66,70. This mechanism with Py proposed the formation of CO2. Thus switched the aerobic surroundings to an anaerobic one. The back formation of the activator requires an additional electron transfer step. These discussions are based on a non-sensitized photo-ATRP system with CuBr2/L. The experimental conditions of this report used lignin as a sensitizer. Here, photoexcited lignin (AL*) and/or oxidized lignin, the polaron formed AL+•, may additionally react with Py, leading to the additional formation of CO2 by the reaction between both. This appears much more complex, requiring complementary investigations to confirm the reaction proposed, which can be time-resolved spectroscopy, optical absorption, or paramagnetic detection of species formed. Such studies would give more insight since the oxygen scrubbing was studied with a non-sensitized system. Although such sensitized systems appear very practical, there would be more fundamental research required to bring them to practical applications. Furthermore, experiments pursued with CuCl2 instead of CuBr2 showed no polymer formation under aerobic conditions.
Practicability – scale-up of photo-ATRP
Figure 7a shows the scale-up experimental device. A scale-up to 100 mL resulted in 15.3 g of PMMA, corresponding to a gravimetric yield of 65% see Fig. 7b. GPC resulted in moderately lower \({\bar{M}}_{{{\rm{n}}}}\) but slightly higher Đ compared with the experimental condition in run 14 (Fig. 7c). The photo-ATRP depicted here used a batch reactor, whose size was adjusted according to the scale-up factor of 25. Larger thickness in the reactor changes absorption conditions and, therefore, polymer radical concentration. This may cause slightly increased termination. Nevertheless, it confirmed that the photo-ATRP initiated by lignin can be scaled up, which provides a promising potential for photo-ATRP sensitized by biomass. More importantly, as a renewable biomass resource, lignin is not only non-toxic but also possesses great advantages in terms of cost. Figure 7d exhibits a comparison of costs for lignin as a heterogeneous photosensitizer, which appeared as at least one-tenth compared to that of other photosensitizers.
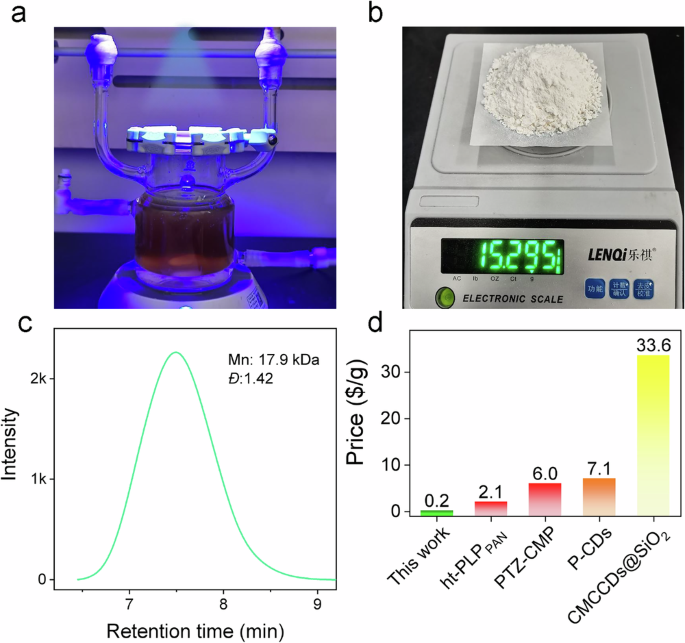
a Diagram of an experimental device for scale-up photo-ATRP; b Photograph of PMMA produced from scale-up photo-ATRP; c GPC of PMMA from scale-up photo-ATRP; d Cost comparison of lignin with other photosensitizers; (ht-PLPPAN = polyacrylonitrile-derived photoactive polymers71, PTZ-CMP: Conjugated cross-linked phenothiazines59, P-CDs: organic network of carbon dots crosslinked with porphyrin53, CMCCDs@SiO2 = carboxymethyl cellulose-based CDs confined in SiO2 matrix52). Prices of other photosensitizers just correspond to the cost of raw materials available from the fine chemical supplier Aladdin. The yield for CDs was calculated as 100%.
link





.jpg)